303232
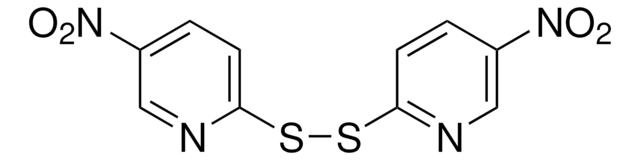
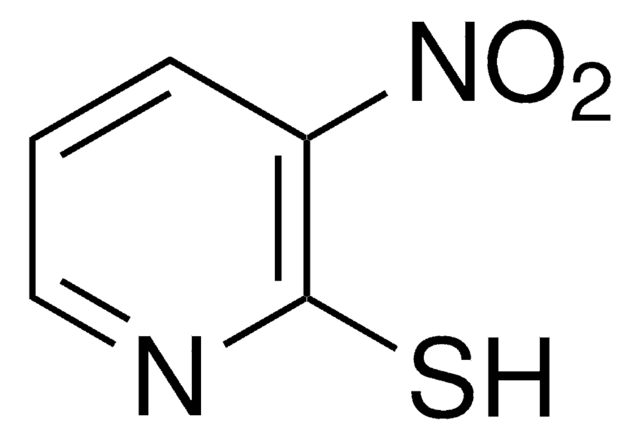
3-Nitro-2-pyridinesulfenyl chloride
95%
Synonym(s):
(3-Nitro-2-pyridyl)sulfenyl chloride, 2-(Chlorosulfanyl)-3-nitropyridine, 3-Nitropyridinyl-2-sulfenyl chloride, [(3-Nitropyridin-2-yl)sulfanyl]chlorane
Sign Into View Organizational & Contract Pricing
All Photos(2)
About This Item
Empirical Formula (Hill Notation):
C5H3ClN2O2S
CAS Number:
Molecular Weight:
190.61
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
95%
mp
205 °C (dec.) (lit.)
solubility
dichloromethane: soluble(lit.)
functional group
nitro
storage temp.
2-8°C
SMILES string
[O-][N+](=O)c1cccnc1SCl
InChI
1S/C5H3ClN2O2S/c6-11-5-4(8(9)10)2-1-3-7-5/h1-3H
InChI key
WTKQMHWYSBWUBE-UHFFFAOYSA-N
General description
The 3-nitro-2-pyridinesulphenyl (Npys) moiety is useful as a protecting-activating group for cysteine, particularly in the synthesis of cyclic and unsymmetrical disulfides. The stability of NpysCl was studied in various solvents.
Application
3-Nitro-2-pyridinesulfenyl chloride (NpysCl) was employed as the starting material for the synthesis of N-, O- and S-Npys-protected amino acid.
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Dam. 1 - Skin Corr. 1B
Storage Class Code
8A - Combustible corrosive hazardous materials
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
R Matsueda et al.
Peptide research, 5(5), 262-264 (1992-09-01)
Two recent reports on the partial lability of the 3-nitro-2-pyridinesulfenyl (Npys) thiol protecting group towards 1-hydroxy-benzotriazole (HOBt) have prompted a rechecking of the chemical behavior of this group. Using both soluble and polymer-bound forms of Cys(Npys) as test materials, the
O Rosen et al.
International journal of peptide and protein research, 35(6), 545-549 (1990-06-01)
The hydroxylic side-chain functional groups of serine, threonine, hydroxproline and tyrosine, the alpha and epsilon-amino moieties of lysine and the thiol group of cysteine were masked by the 3-nitro-2-pyridinesulfenyl (Npys) protecting group. Deprotection was mildly affected by thiolysis with either
S Rajagopalan et al.
International journal of peptide and protein research, 45(2), 173-179 (1995-02-01)
TASPs (template-assembled synthetic peptides) are generated by the covalent attachment of linear peptides to a common peptide backbone, thus generating larger synthetic peptides/proteins with prefolded structure. In this work we present a strategy for the synthesis of a heterotemplate-assembled synthetic
R G Simmonds et al.
International journal of peptide and protein research, 43(4), 363-366 (1994-04-01)
The 3-nitro-2-pyridinesulphenyl (Npys) moiety is finding increasing utility as a protecting-activating group for cysteine, particularly in the synthesis of cyclic and unsymmetrical disulfides using the Boc strategy. This chemistry has been extended to peptides assembled by the Fmoc strategy. N-Terminal
K C Pugh et al.
International journal of peptide and protein research, 42(2), 159-164 (1993-08-01)
3-Nitro-2-pyridinesulfenyl chloride (NpysCl) is the starting material for the synthesis of N-, O- and S-Npys-protected amino acids. Two efficient, novel synthetic routes to NpysCl are described. The stability of NpysCl was determined in a variety of solvents, with and without
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service