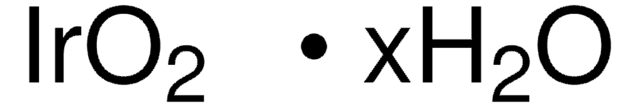208833
Ruthenium(IV) oxide hydrate
powder
Synonym(s):
Hydrous ruthenium oxide, Ruthenium dioxide hydrate
Sign Into View Organizational & Contract Pricing
All Photos(4)
About This Item
Linear Formula:
RuO2 · xH2O
CAS Number:
Molecular Weight:
133.07 (anhydrous basis)
EC Number:
MDL number:
UNSPSC Code:
12352303
PubChem Substance ID:
NACRES:
NA.23
Recommended Products
form
powder
reaction suitability
reagent type: catalyst
core: ruthenium
SMILES string
O.O=[Ru]=O
InChI
1S/H2O.2O.Ru/h1H2;;;
InChI key
FGEKTVAHFDQHBU-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Hui Wang et al.
Journal of hazardous materials, 154(1-3), 44-50 (2007-11-13)
By using a self-made carbon/polytetrafluoroethylene (C/PTFE) O2-fed as the cathode and Ti/IrO2/RuO2 as the anode, the degradation of three organic compounds (phenol, 4-chlorophenol, and 2,4-dichlorophenol) was investigated in the diaphragm (with terylene as diaphragm material) electrolysis device by electrochemical oxidation
Aleksandar R Zeradjanin et al.
ChemSusChem, 5(10), 1897-1904 (2012-08-16)
The reaction path of the Cl(2) evolution reaction (CER) was investigated by combining electrochemical and spectroscopic methods. It is shown that oxidation and reconstruction of the catalyst surface during CER is a consequence of the interaction between RuO(2) and water.
M Gonçalves et al.
Bioresource technology, 99(17), 8207-8211 (2008-05-02)
Electrochemical treatment of oleate using RuO2 and IrO2 type dimensionally stable anodes in alkaline medium was performed to develop a feasible anaerobic pre-treatment of fatty effluents. The results showed that the pre-treated solutions over RuO2 were faster degraded by anaerobic
Syaifullah Muhammad et al.
Journal of hazardous materials, 215-216, 183-190 (2012-03-16)
Activated carbon (AC) and Zeolite Socony Mobil-5 (ZSM5) supported ruthenium oxide catalysts were prepared and tested to degrade aqueous phenol in the presence of peroxymonosulphate. The physicochemical properties of ruthenium oxide based catalysts were characterised by several techniques such as
Serge Zhuiykov et al.
Talanta, 82(2), 502-507 (2010-07-07)
A Cu(2)O-doped RuO(2) sensing electrode (SE) for potentiometric detection of dissolved oxygen (DO) was prepared and its structure and electrochemical properties were analyzed by scanning electron microscopy (SEM), X-ray diffraction (XRD), X-ray photoelectron microscopy (XPS) and energy-dispersive spectroscopy (EDS) techniques.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service