E4402
2-Ethoxybenzamid
97%
Anmeldenzur Ansicht organisationsspezifischer und vertraglich vereinbarter Preise
Alle Fotos(1)
About This Item
Lineare Formel:
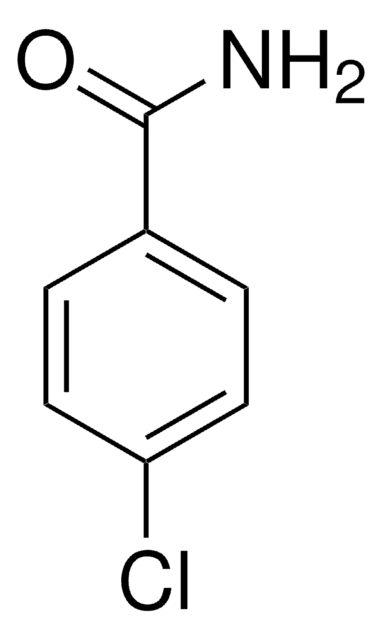
C2H5OC6H4CONH2
CAS-Nummer:
Molekulargewicht:
165.19
EG-Nummer:
MDL-Nummer:
UNSPSC-Code:
12352100
PubChem Substanz-ID:
NACRES:
NA.22
Empfohlene Produkte
Assay
97%
mp (Schmelzpunkt)
132-134 °C (lit.)
SMILES String
CCOc1ccccc1C(N)=O
InChI
1S/C9H11NO2/c1-2-12-8-6-4-3-5-7(8)9(10)11/h3-6H,2H2,1H3,(H2,10,11)
InChIKey
SBNKFTQSBPKMBZ-UHFFFAOYSA-N
Verwandte Kategorien
Lagerklassenschlüssel
11 - Combustible Solids
WGK
WGK 2
Flammpunkt (°F)
Not applicable
Flammpunkt (°C)
Not applicable
Persönliche Schutzausrüstung
Eyeshields, Gloves, type N95 (US)
Analysenzertifikate (COA)
Suchen Sie nach Analysenzertifikate (COA), indem Sie die Lot-/Chargennummer des Produkts eingeben. Lot- und Chargennummern sind auf dem Produktetikett hinter den Wörtern ‘Lot’ oder ‘Batch’ (Lot oder Charge) zu finden.
Besitzen Sie dieses Produkt bereits?
In der Dokumentenbibliothek finden Sie die Dokumentation zu den Produkten, die Sie kürzlich erworben haben.
Yoshihiro Hayashi et al.
Pharmaceutics, 12(7) (2020-07-02)
We previously reported a novel method for the precise prediction of tablet properties (e.g., tensile strength (TS)) using a small number of experimental data. The key technique of this method is to compensate for the lack of experimental data by
N Hirasawa et al.
Chemical & pharmaceutical bulletin, 47(3), 417-420 (1999-04-23)
Solid dispersions of carbamazepine or ethenzamide were prepared by melting and rapid cooling with liquid nitrogen using lactose as a carrier. The physical characteristics of these solid dispersions were investigated by powder X-ray diffraction, differential scanning calorimetry, and dissolution rate
Y Miyamoto et al.
Chemical & pharmaceutical bulletin, 46(9), 1432-1437 (1998-10-17)
A computer optimization technique based on surface response methodology was applied to optimize the wet granulation process for designing tablets. Physical properties (mean granule size, granule size distribution, compressibility, granule strength) of a model granule formulation containing ethenzamide were accurately
Tadashi Fukunaka et al.
Journal of pharmaceutical sciences, 94(5), 1004-1012 (2005-03-29)
Milling is a common procedure to improve bioavailability of many active pharmaceutical ingredients (APIs), which typically have low solubility in water. But such micronization can yield an increase in the cohesiveness of particles. Although particle cohesiveness is desirable for tablet
H Uehara et al.
Cancer letters, 135(1), 83-90 (1999-03-17)
Six-week-old male F344 rats were given a mixture of 0.01% diethylnitrosamine, 0.05% N-butyl-N-(4-hydroxybutyl)nitrosamine and 0.02% N-methyl-N'-nitro-N-nitrosoguanidine in their drinking water for 1 week. When 0.8%, 0.4%, or 0% of a mixture of non-steroidal anti-inflammatory drugs (NSAIDs) (acetaminophen, aspirin, dipyrone plus
Unser Team von Wissenschaftlern verfügt über Erfahrung in allen Forschungsbereichen einschließlich Life Science, Materialwissenschaften, chemischer Synthese, Chromatographie, Analytik und vielen mehr..
Setzen Sie sich mit dem technischen Dienst in Verbindung.