Select a Size
| Size/SKU | Availability | Price |
|---|---|---|
200 mg | Please contact Customer Service for Availability | 10 900,00 CZK |
About This Item
grade
pharmaceutical primary standard
agency
USP
API family
pentoxifylline
manufacturer/tradename
USP
application(s)
pharmaceutical (small molecule)
format
neat
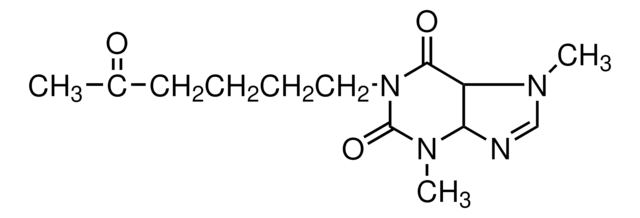
SMILES string
CN1C=NC2C1C(=O)N(CCCCC(C)=O)C(=O)N2C
InChI
1S/C13H20N4O3/c1-9(18)6-4-5-7-17-12(19)10-11(14-8-15(10)2)16(3)13(17)20/h8,10-11H,4-7H2,1-3H3
InChI key
MQGNNJQTNFHNHK-UHFFFAOYSA-N
General description
Application
- Pentoxifylline Compounded Oral Suspension
- Pentoxifylline Extended-Release Tablets
Biochem/physiol Actions
Analysis Note
Other Notes
1 of 1
This Item | |||
|---|---|---|---|
| manufacturer/tradename USP | manufacturer/tradename EDQM | manufacturer/tradename - | manufacturer/tradename - |
| grade pharmaceutical primary standard | grade pharmaceutical primary standard | grade certified reference material, pharmaceutical secondary standard | grade - |
| format neat | format neat | format - | format - |
| application(s) pharmaceutical (small molecule) | application(s) pharmaceutical (small molecule) | application(s) pharmaceutical | application(s) - |
| API family pentoxifylline | API family pentoxifylline | API family pentoxifylline | API family - |
Still not finding the right product?
Explore all of our products under Pentoxifylline
signalword
Warning
hcodes
Hazard Classifications
Acute Tox. 4 Oral - Lact.
Storage Class
11 - Combustible Solids
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.