Select a Size
| Size/SKU | Availability | Price |
|---|---|---|
2 mg | Please contact Customer Service for Availability | CZK 1,730.00 |
10 mg | Please contact Customer Service for Availability | CZK 4,200.00 |
25 mg | Please contact Customer Service for Availability | CZK 8,270.00 |
50 mg | Please contact Customer Service for Availability | CZK 23,300.00 |
About This Item
CZK 1,730.00
biological source
synthetic (organic)
Quality Segment
assay
≥95% (HPLC)
form
powder
mol wt
~_1.0 kDa
color
white to yellow-white
solubility
H2O: 50 mg/mL
storage temp.
−20°C
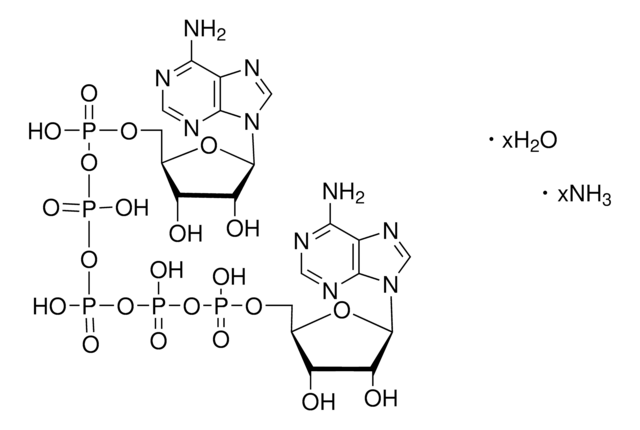
SMILES string
[Na+].[Na+].[Na+].[Na+].[Na+].Nc1ncnc2n(cnc12)[C@@H]3O[C@H](COP([O-])(=O)OP([O-])(=O)OP([O-])(=O)OP([O-])(=O)OP([O-])(=O)OC[C@H]4O[C@H]([C@H](O)[C@@H]4O)n5cnc6c(N)ncnc56)[C@@H](O)[C@H]3O
InChI
1S/C20H29N10O22P5.5Na/c21-15-9-17(25-3-23-15)29(5-27-9)19-13(33)11(31)7(47-19)1-45-53(35,36)49-55(39,40)51-57(43,44)52-56(41,42)50-54(37,38)46-2-8-12(32)14(34)20(48-8)30-6-28-10-16(22)24-4-26-18(10)30;;;;;/h3-8,11-14,19-20,31-34H,1-2H2,(H,35,36)(H,37,38)(H,39,40)(H,41,42)(H,43,44)(H2,21,23,25)(H2,22,24,26);;;;;/q;5*+1/p-5/t7-,8-,11-,12-,13-,14-,19-,20-;;;;;/m1...../s1
InChI key
NNMFUJJMJIYTSP-CSMIRWGRSA-I
General description
Application
- an adenylate kinase (AK) inhibitor in: sarcoma osteogenic (Saos-2) cells
- mitochondrial lysates during ATP synthesis
- tetramethylrhodamine methyl ester (TMRM) based membrane potential assay
- chromoplasts
Biochem/physiol Actions
1 of 1
This Item | |||
|---|---|---|---|
| biological source synthetic (organic) | biological source synthetic (organic) | biological source synthetic (organic) | biological source synthetic (inorganic) |
| assay ≥95% (HPLC) | assay ≥95% (HPLC) | assay ≥95% | assay ≥96% |
| Quality Level 200 | Quality Level 200 | Quality Level 200 | Quality Level 200 |
| form powder | form powder | form powder | form powder |
| solubility H2O: 50 mg/mL | solubility H2O: 50 mg/mL | solubility H2O: 50 mg/mL | solubility water: 25 mg/mL, clear, colorless to very faintly yellow |
| storage temp. −20°C | storage temp. −20°C | storage temp. −20°C | storage temp. −20°C |
Storage Class
11 - Combustible Solids
wgk
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Gloves, type N95 (US)
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.