Select a Size
| Pack Size | SKU | Availability | Price |
|---|---|---|---|
| 1 mg | Please contact Customer Service for Availability | CZK 5,630.00 |
About This Item
Quality Level
assay
≥95%
form
powder
UniProt accession no.
storage temp.
−20°C
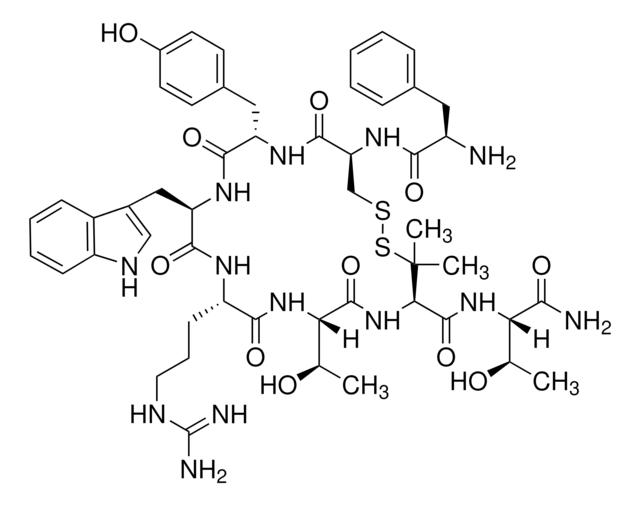
SMILES string
C[C@@H](O)[C@H](NC(=O)[C@H]1NC(=O)[C@@H](NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@@H](Cc2c[nH]c3ccccc23)NC(=O)[C@H](Cc4ccc(O)cc4)NC(=O)[C@H](CSSC1(C)C)NC(=O)[C@H](N)Cc5ccccc5)[C@@H](C)O)C(N)=O
InChI
1S/C51H69N13O11S2/c1-26(65)39(42(53)68)62-49(75)41-51(3,4)77-76-25-38(61-43(69)33(52)21-28-11-6-5-7-12-28)47(73)59-36(22-29-16-18-31(67)19-17-29)45(71)60-37(23-30-24-57-34-14-9-8-13-32(30)34)46(72)58-35(15-10-20-56-50(54)55)44(70)63-40(27(2)66)48(74)64-41/h5-9,11-14,16-19,24,26-27,33,35-41,57,65-67H,10,15,20-23,25,52H2,1-4H3,(H2,53,68)(H,58,72)(H,59,73)(H,60,71)(H,61,69)(H,62,75)(H,63,70)(H,64,74)(H4,54,55,56)/t26-,27-,33-,35+,36+,37-,38+,39+,40+,41-/m1/s1
InChI key
OFMQLVRLOGHAJI-FGHAYEPSSA-N
Gene Information
rat ... Pnoc(25516)
Looking for similar products? Visit Product Comparison Guide
1 of 1
This Item | |||
|---|---|---|---|
| assay ≥95% | assay ≥97% (HPLC) | assay - | assay - |
| Quality Level 200 | Quality Level 100 | Quality Level 100 | Quality Level 100 |
| form powder | form powder | form lyophilized powder | form lyophilized powder |
| UniProt accession no. | UniProt accession no. | UniProt accession no. - | UniProt accession no. - |
| Gene Information rat ... Pnoc(25516) | Gene Information mouse ... Pnoc(18155) | Gene Information mouse ... PNO1(66249), Pno1(66249) | Gene Information mouse ... SLC20A2(20516), Slc20a2(20516) |
| storage temp. −20°C | storage temp. −20°C | storage temp. −20°C | storage temp. −20°C |
Application
- to study the anti-hyperalgesic effect of dipeptidyl peptidase 4 (DPP4) inhibitor isoleucine-proline-isoleucine (IPI) and vildagliptin in carrageenan-induced inflammation
- to study the role of MOR in glutamate and gamma-aminobutyric acid (GABA) efflux during predator stress in rats[1]
- to determine the endogenous opioid peptide involved in blocking pain induced by activated gastrin-releasing peptide (Grp+) neurons
Biochem/physiol Actions
Storage Class
11 - Combustible Solids
wgk
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Gloves, type N95 (US)
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Global Trade Item Number
| SKU | GTIN |
|---|---|
| C6352-1MG | 04061826152607 |
Active Filters
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service