Sign In to View Organizational & Contract Pricing.
Select a Size
Change View
| Size/SKU | Availability | Price |
|---|---|---|
5 mg | Please contact Customer Service for Availability | CZK 4,920.00 |
About This Item
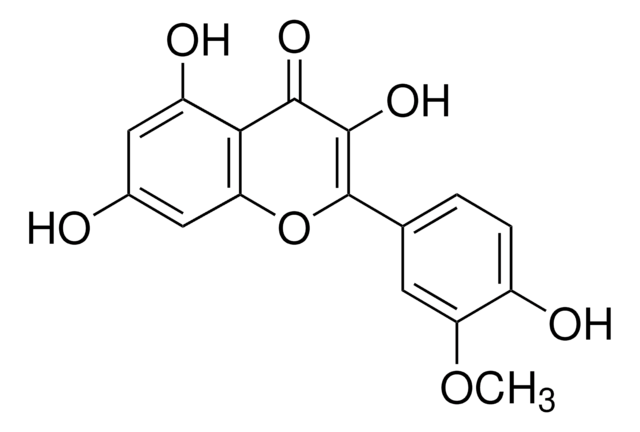
Empirical Formula (Hill Notation):
C16H12O7
CAS Number:
Molecular Weight:
316.26
UNSPSC Code:
12352205
NACRES:
NA.47
PubChem Substance ID:
EC Number:
207-545-5
Beilstein/REAXYS Number:
44723
MDL number:
Quality Segment
assay
≥95.0% (HPLC)
application(s)
metabolomics
vitamins, nutraceuticals, and natural products
SMILES string
COc1cc(ccc1O)C2=C(O)C(=O)c3c(O)cc(O)cc3O2
InChI
1S/C16H12O7/c1-22-11-4-7(2-3-9(11)18)16-15(21)14(20)13-10(19)5-8(17)6-12(13)23-16/h2-6,17-19,21H,1H3
InChI key
IZQSVPBOUDKVDZ-UHFFFAOYSA-N
General description
Isorhamnetin is an O-methylated flavonol [1] found in fruits, plants, and medicinal herbs. It is a metabolite of quercetin.
Application
Isorhamnetin (IRN) has been used:
- as a neuroprotective agent to test its effect on scopolamine-induced cortico-hippocampal learning and memory deficiency in mice[2]
- as a reference standard in quadrupole time-of-flight mass spectrometry (QTOF MS) to analyze and quantify the phenolic compounds present in honey extract[3]
- as a reference standard to determine the phenolic profile of Artemisia species using Reversed-phase high-performance liquid chromatography with diode array detection and electrospray ionization mass spectrometry (RP-HPLC-DAD-ESI-TQ-MS/MS)[4]
Biochem/physiol Actions
Isorhamnetin exerts antioxidant, anti-inflammatory, anti-cancer, anti-bacterial, and anti-viral effects.[5] It has shown to be a potential neuroprotective agent by inhibiting neurodegeneration in the hippocampus under Diabetes mellitus (DM) conditions.[1] Isorhamnetin at low levels may serve as an anti-diabetic agent by promoting glucose uptake in skeletal muscle cells and maintaining glucose homeostasis in hyperglycemic conditions. It regulates cell signaling pathways such as Nuclear factor kappa B (NF-κB), phosphatidylinositol 3-kinase (PI3K)/protein kinase B (AKT), mitogen-activated protein kinase (MAPK) etc.[5] Isorhamnetin elicits protective and therapeutic effects on atherosclerosis and cardiovascular diseases.[5]
Isorhamnetin inhibits adipogenesis by interfering with differentiation of adipose stem cells, by a mechanism involving stabilization of β-catenin and up-regulating the Wnt signaling pathway.
Packaging
Bottomless glass bottle. Contents are inside inserted fused cone.
1 of 1
This Item | |||
|---|---|---|---|
| assay ≥95.0% (HPLC) | assay ≥99% (HPLC) | assay ≥95.0% (HPLC) | assay ≥95.0% (HPLC) |
| application(s) metabolomics | application(s) food and beverages | application(s) - | application(s) food and beverages |
| Quality Level 100 | Quality Level 100 | Quality Level - | Quality Level - |
Still not finding the right product?
Explore all of our products under Isorhamnetin
Storage Class
11 - Combustible Solids
wgk
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Gloves, type P3 (EN 143) respirator cartridges
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.