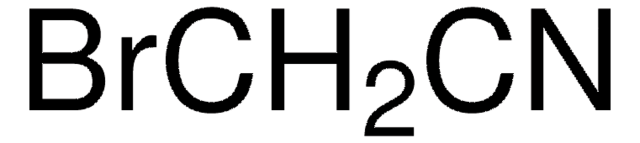All Photos(1)
About This Item
Linear Formula:
C6H5OCH2CN
CAS Number:
Molecular Weight:
133.15
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
98%
refractive index
n20/D 1.524 (lit.)
bp
235-238 °C (lit.)
density
1.09 g/mL at 25 °C (lit.)
SMILES string
N#CCOc1ccccc1
InChI
1S/C8H7NO/c9-6-7-10-8-4-2-1-3-5-8/h1-5H,7H2
InChI key
VLLSCJFPVSQXDM-UHFFFAOYSA-N
Application
Phenoxyacetonitrile may be used in the synthesis of:
- 2,4-dihydroxyphenoxyacetophenones
- methylthio(phenoxy)acetonitrile
- 2,4-diamino-5-(3,4,5-trimethoxyphenoxy)pyrimidine
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 4 Dermal - Acute Tox. 4 Inhalation - Acute Tox. 4 Oral
Storage Class Code
10 - Combustible liquids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Reactions of quinoline and 4-chloroquinoline 1-oxides with phenoxyacetonitrile, chloromethylphenylsulfone, and methyl-thiomethyl-p-tolylsulfone.
Hamana M, et al.
Heterocycles, 25, 229-233 (1987)
Chemistry of modified flavonoids.
Arkhipov VV, et al.
Chemistry of Heterocyclic Compounds, 33(5), 515-519 (1997)
A Simple Procedure for the Preparation of (Methyl-and Phenylthio) aryloxyacetonitriles.
Jonczyk A and Muttar EH
Organic preparations and procedures international, 25(6), 690-693 (1993)
Synthesis of trimethoprim variations. Replacement of methylene by polar groupings.
Stogryn EL.
Journal of Medicinal Chemistry, 15(2), 200-201 (1972)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service