All Photos(3)
About This Item
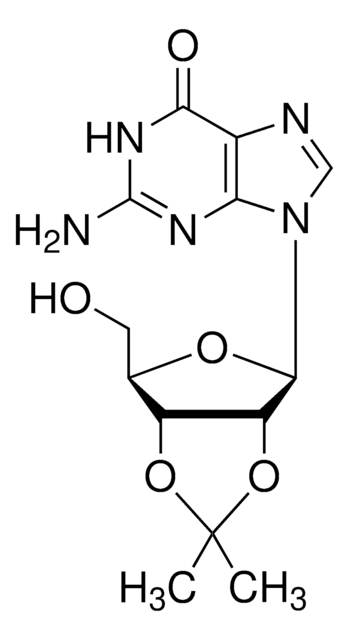
Empirical Formula (Hill Notation):
C12H16N2O6
CAS Number:
Molecular Weight:
284.27
EC Number:
MDL number:
UNSPSC Code:
41106305
PubChem Substance ID:
NACRES:
NA.51
Recommended Products
biological source
synthetic (organic)
Quality Level
Assay
≥99% (HPLC)
form
powder
solubility
water: 50 mg/mL, clear to very slightly hazy, colorless to light yellow
storage temp.
−20°C
SMILES string
CC1(C)O[C@@H]2[C@@H](CO)O[C@H]([C@@H]2O1)N3C=CC(=O)NC3=O
InChI
1S/C12H16N2O6/c1-12(2)19-8-6(5-15)18-10(9(8)20-12)14-4-3-7(16)13-11(14)17/h3-4,6,8-10,15H,5H2,1-2H3,(H,13,16,17)/t6-,8-,9-,10-/m1/s1
InChI key
GFDUSNQQMOENLR-PEBGCTIMSA-N
Looking for similar products? Visit Product Comparison Guide
Application
2′,3′-O-Isopropylideneuridine is used in the chemical synthesis of N-benzoylated uridine derivatives and N3-substituted 2′,3′-O-isopropylideneuridines with central nervous system (CNS) depressant activity.
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Tomomi Shimizu et al.
Chemical & pharmaceutical bulletin, 53(3), 313-318 (2005-03-04)
Seventy eight N(3)-substituted derivatives of uridine (1), thymidine (2), 2'-deoxyuridine (3), 6-azauridine (4), 2',3'-O-isopropylideneuridine (5), and arabinofuranosyluracil (6) were synthesized and their antinociceptive effects were evaluated. N(3)-(2',4'-Dimethoxyphenacyl)uridine (1l), N(3)-(2',4'-dimethoxyphenacyl)2'-deoxyuridine (3l), and N(3)-(2',5'-dimethoxyphenacyl)arabinofuranosyluracil (6m) possessed 93, 86, and 82% of the
C S Yao et al.
Chemical & pharmaceutical bulletin, 47(12), 1802-1804 (2000-04-05)
N3-Substituted derivatives of 2',3'-O-isopropylideneuridine (1) were synthesized and their pharmacological effects on the central nervous system (CNS) examined using mice. Methyl (2), ethyl (3), propyl (4), butyl (5), allyl (6), benzyl (7), o-, m-, p-xylyls (8, 9, 10), and alpha-phenylethyl
M L Scholes et al.
International journal of radiation biology, 61(4), 443-449 (1992-04-01)
The effect of pH on base release in the gamma-radiolysis of N2O-saturated solutions of a number of nucleosides (including uridine, 3-methyluridine, 2',3'-O-isopropylidene-uridine, and adenosine) has been investigated. For all these nucleotides, independent of the base or sugar moiety, base release
S I Malekin et al.
Bioorganicheskaia khimiia, 23(8), 648-654 (1997-08-01)
New nucleoside-phospholipid conjugates were synthesized based on 1,2-disubstituted glycerides and nucleosides. These contain rac-1-hexadecyl-2-palmitoyl(or 2-methylcarbamoyl)-sn-glycero-3-phosphate as the phospholipid component and 2',3'-didehydro-3'-deoxythymidine, 1-(Z-5-hydroxypentene-2-yl)thymine, or 2',3'-isopropylideneuridine as a nucleoside component. The conjugates were synthesized by three different ways: from rac-1-hexadecyl-2-acyl-sn-glycero-3-phospodichlorides, -3-phosphatidic acids
Anita R Maguire et al.
Carbohydrate research, 337(4), 369-372 (2002-02-14)
An improved procedure for the synthesis of N-benzoyl-2',3'-O-isopropylidene uridine via one-step selective N-benzoylation of 2',3' -O-isopropylidene uridine has been developed. An efficient synthetic route to N-benzoyl thymidine via initial tribenzoylation, followed by selective hydrolysis of the benzoates is also described.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service