S7556
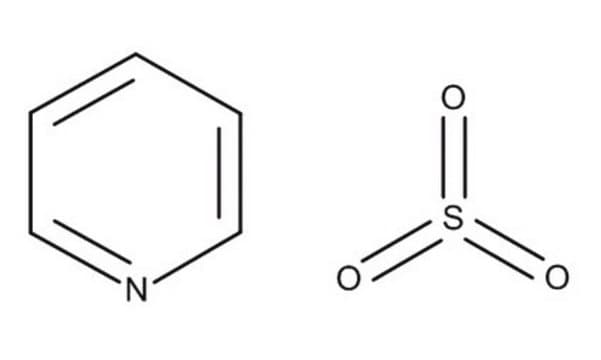
Sulfur trioxide pyridine complex
97%
Synonym(s):
NSC 75831, Pyridine sulfur trioxide complex
Sign Into View Organizational & Contract Pricing
All Photos(4)
About This Item
Empirical Formula (Hill Notation):
C5H5NO3S
CAS Number:
Molecular Weight:
159.16
Beilstein:
3704116
EC Number:
MDL number:
UNSPSC Code:
12352303
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
97%
reaction suitability
reagent type: oxidant
SMILES string
O=S(=O)=O.c1ccncc1
InChI
1S/C5H5N.O3S/c1-2-4-6-5-3-1;1-4(2)3/h1-5H;
InChI key
UDYFLDICVHJSOY-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
Application
- Sulfation agent
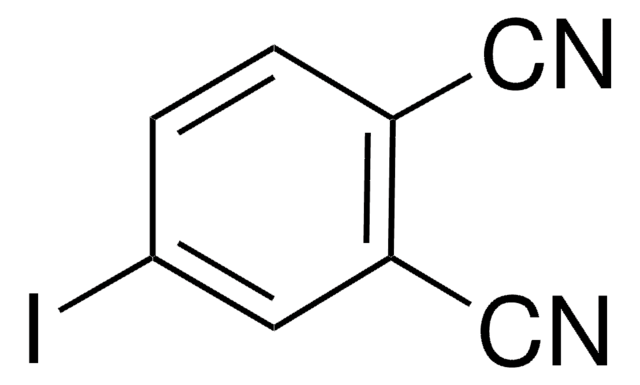
Reactant for:
- Preparation of azido anologs of pregnanolone
- Sulfate esters of morphine derivatives
- Gold nanoparticles capped with sulfate-ended ligands as anti-HIV agents
Sulfur trioxide-pyridine (SO3-py) complex can be used to convert the allylic or benzylic hydroxyl group of compounds such as geraniol, farnesol, and 1-indanol into sulfate monoester, which is a good leaving group. This method is highly useful in the total synthesis of sesquiterpenes.
As a sulfating agent, SO3-py complex can be used for the preparation of sulfated-chitins, sulfated hyaluronan (HyaS) derivatives and sulfated starch.
As a sulfating agent, SO3-py complex can be used for the preparation of sulfated-chitins, sulfated hyaluronan (HyaS) derivatives and sulfated starch.
related product
Product No.
Description
Pricing
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 4 Oral - Eye Dam. 1 - Skin Corr. 1B
Storage Class Code
8A - Combustible corrosive hazardous materials
WGK
WGK 3
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Polysaccharide-based superporous hydrogels with fast swelling and superabsorbent properties.
Kuang J, et al.
Carbohydrate Polymers, 83(1), 284-290 (2011)
A method for deoxygenation of allylic and benzylic alcohols.
Corey E J and Achiwa K
The Journal of Organic Chemistry, 34(11), 3667-3668 (1969)
Preparation of sulfated-chitins under homogeneous conditions.
Zou Y and Khor E
Carbohydrate Polymers, 77(3), 516-525 (2009)
Sulfated hyaluronan derivatives reduce the proliferation rate of primary rat calvarial osteoblasts.
Kunze R, et al.
Glycoconjugate Journal, 27(1), 151-158 (2010)
Helyn Priscila de Oliveira Barddal et al.
International journal of biological macromolecules, 145, 604-610 (2019-12-31)
Heparin is an extremely important and recognized anticoagulant and antithrombotic agent. Obtained from animal sources and being highly potent, risks of contamination by pathogens and bleeding are some concerns related to heparin use. In the search for alternatives to heparin
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service



![1,8-Diazabicyclo[5.4.0]undec-7-ene 98%](/deepweb/assets/sigmaaldrich/product/structures/120/564/5b373e23-1624-489c-8efb-692de0f96ffb/640/5b373e23-1624-489c-8efb-692de0f96ffb.png)

![1,5,7-Triazabicyclo[4.4.0]dec-5-ene 98%](/deepweb/assets/sigmaaldrich/product/structures/171/446/333d560c-cff6-4958-b489-5acfb3057cce/640/333d560c-cff6-4958-b489-5acfb3057cce.png)