B75956
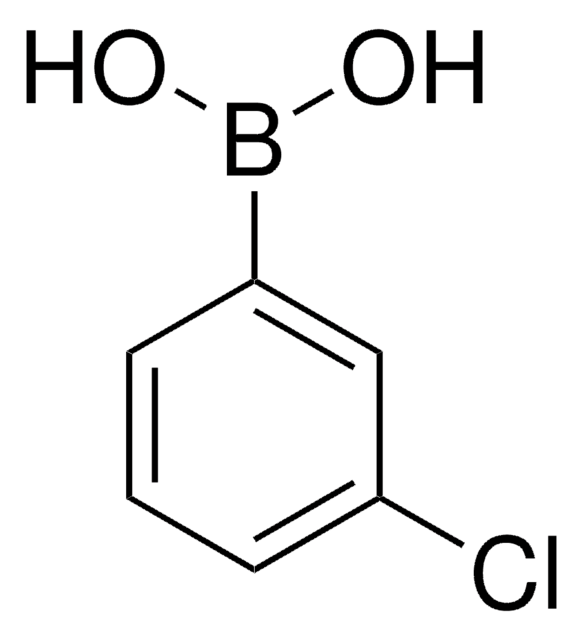
4-Bromophenylboronic acid
≥95.0%
Synonym(s):
(p-Bromophenyl)boronic acid, 4-Bromobenzeneboronic acid, 4-Bromophenylboric acid, p-Bromobenzeneboronic acid, p-Bromophenylboric acid, NSC 25407
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Linear Formula:
BrC6H4B(OH)2
CAS Number:
Molecular Weight:
200.83
Beilstein:
2936347
EC Number:
MDL number:
UNSPSC Code:
12352103
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
≥95.0%
95%
form
crystals
mp
284-288 °C (lit.)
SMILES string
OB(O)c1ccc(Br)cc1
InChI
1S/C6H6BBrO2/c8-6-3-1-5(2-4-6)7(9)10/h1-4,9-10H
InChI key
QBLFZIBJXUQVRF-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Application
Reagent used for
Reagent used in Preparation of
- Palladium catalyzed Suzuki-Miyaura cross-couplings
- Pd(II)-catalyzed diastereoselective conjugate additions
- Palladium-catalyzed stereoselective Heck-type reaction of allylic esters with arylboronic acids
- Tandem-type Pd(II)-catalyzed oxidative Heck reaction and intramolecular C-H amidation sequence
- Copper-mediated ligandless aerobic fluoroalkylation of arylboronic acids with fluoroalkyl iodides
- Pd-catalyzed arylative cyclization of alkyne-tethered enals or enones via carbopalladation of alkynes
- Copper-catalyzed cross-couplings
Reagent used in Preparation of
- Gallate-based obovatol analogs with potential anti-tumor activity
- Protein modulators and enzymatic and kinase inhibitors
Other Notes
Contains varying amounts of anhydride
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Xing Li et al.
Organic & biomolecular chemistry, 10(3), 495-497 (2011-11-22)
Suzuki-Miyaura cross-couplings of arenediazonium salts with arylboronic acids catalyzed by highly active aluminium hydroxide-supported palladium nanoparticles catalyst have been investigated for the first time. The reactions are performed at 25 °C in MeOH without any base and ligand to afford
Erin Tay et al.
Pharmaceutics, 12(1) (2019-12-28)
Lipid based formulations (LBFs) are commonly employed to enhance the absorption of highly lipophilic, poorly water-soluble drugs. However, the utility of LBFs can be limited by low drug solubility in the formulation. Isolation of ionizable drugs as low melting, lipophilic
Chuan Xiao et al.
Bioorganic & medicinal chemistry, 19(23), 7100-7110 (2011-11-01)
A series of purine nucleoside analogues bearing an aryl and hetaryl group in position 6 were prepared and their biological activities were assessed by in vitro CDK1/Cyclin B1 and CDK2/Cyclin A2 kinase assay. From the synthesized chemicals, three Xylocydine derivatives
Anna C Pham et al.
Journal of controlled release : official journal of the Controlled Release Society, 268, 400-406 (2017-11-04)
Phosphorylated tocopherols are a new class of lipid excipients that have demonstrated potential in pharmaceutical applications. Their ability to solubilise poorly water soluble drugs indicates their potential utility in improving bioavailability of drugs where solubility limits their bioavailability. In this
Synthesis of obovatol derivatives and their preliminary evaluation as antitumor agents
Lee, M.-S.; et al.
Bull. Korean Chem. Soc., 28, 1601-1604 (2007)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service