860166
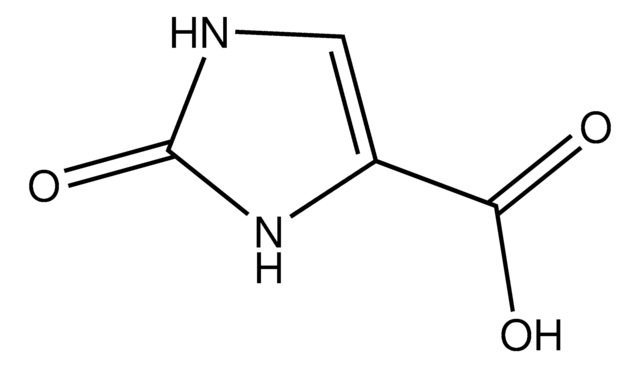
2-Imidazolidone-4-carboxylic acid
95%
Synonym(s):
2-Oxo-4-imidazolidinecarboxylic acid
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Empirical Formula (Hill Notation):
C4H6N2O3
CAS Number:
Molecular Weight:
130.10
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
95%
form
solid
mp
184-186 °C (dec.) (lit.)
functional group
carboxylic acid
SMILES string
OC(=O)C1CNC(=O)N1
InChI
1S/C4H6N2O3/c7-3(8)2-1-5-4(9)6-2/h2H,1H2,(H,7,8)(H2,5,6,9)
InChI key
KZKRPYCBSZIQKN-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Application
Reactant for synthesis of human BACE-1 inhibitors
Additive for crystal nucleation and growth
Additive for crystal nucleation and growth
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
S Samuels et al.
Pharmacology, biochemistry, and behavior, 24(1), 143-146 (1986-01-01)
DBA/2J mice were exposed in utero, between days 15-18 of gestation, to either of two enzyme inhibitors, previously shown to decrease blood-brain, large-neutral amino acid transport in adults: L-methionine-RS-sulfoximine and 2-imidazolidone-4-carboxylic acid. The young mice demonstrated persistently altered motor behavior
Heuisul Park et al.
Bioorganic & medicinal chemistry letters, 18(9), 2900-2904 (2008-04-25)
We describe synthesis and evaluation of a series of cyclic urea derivatives with hydroxylethylamine isostere. Modification of P3, P1, and P2' and combination of SAR display a >100-fold increase in potency with good cellular activity (IC(50)=0.15microM) relative to the previously
S. Devarakonda, et al.,
Journal of Crystal Growth, 204, 525-538 (1999)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service







![2-[2-(2-Aminoethoxy)ethoxy]ethanol ≥96.0% (GC)](/deepweb/assets/sigmaaldrich/product/structures/237/185/b94eadd2-5a2c-4a5b-a13d-3d3905df0dbc/640/b94eadd2-5a2c-4a5b-a13d-3d3905df0dbc.png)
