All Photos(2)
About This Item
Linear Formula:
BrCH2C6H3(NO2)CO2H
CAS Number:
Molecular Weight:
260.04
Beilstein:
1970939
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
97%
form
solid
mp
127-130 °C (lit.)
solubility
DMF: soluble(lit.)
dichloromethane: soluble(lit.)
SMILES string
OC(=O)c1ccc(CBr)c(c1)[N+]([O-])=O
InChI
1S/C8H6BrNO4/c9-4-6-2-1-5(8(11)12)3-7(6)10(13)14/h1-3H,4H2,(H,11,12)
InChI key
QMAHVAFURJBOFV-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
General description
4-Bromomethyl-3-nitrobenzoic acid (BNBA) is a benzoic acid derivative. It has been synthesized by the nitration of 4-bromomethylbenzoic acid using fuming nitric acid. It participates in the synthesis of 3,4-dihydro-2(1H)-quinazolinones and 3,4-dihydro-1H-quinazolin-2-thiones.
Application
4-Bromomethyl-3-nitrobenzoic acid may be used in the following studies:
- As a reactant in the synthesis of 4-bromomethyl-3-nitrobenzoic acid succinimide ester (BNBA-SE).
- As a reactant in the synthesis of 4-(2-hydroxyethylmercaptylmethyl)-3-nitrobenzoic acid.
- As a reactant in the synthesis of decyl 4-(bromomethyl)-3-nitrobenzoate.
- As a reactant in the synthesis of 4-((2-(hydroxymethyl)phenylamino)methyl)-3-nitrobenzoic acid.
- As a thiol photo-deprotection reagent.
- As a UV-cleavable reagent to functionalize polyacrylamide acryl hydrate (PAAH).
- As a photocleavable linker to form localized photoinversion of surface charges inside the colloidal crystal.
- As a starting material in the synthesis of 2H-indazole based library using parallel solution-phase methods.
- As a reactant in the synthesis of (N-allyloxycarbonyl)-5-aminopent-1-yl 4-bromomethyl-3-nitrobenzoate.
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Skin Corr. 1B
Storage Class Code
8A - Combustible corrosive hazardous materials
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Hai-Yuan Hsu et al.
Molecular diversity, 16(2), 241-249 (2011-12-20)
An efficient microwave-assisted and water-soluble ionic liquid (IL)-supported synthesis of medicinally important dihydro- and tetrahydroquinazolines has been developed. The protocol involves the S(N)2 substitution reaction of IL-bound 4-bromomethyl-3-nitrobenzoic acid with various primary amines to provide IL-bound 4-((alkylamino) methyl)-3-nitrobenzoate under microwave
Solid-phase synthesis of 3, 4-dihydro-2 (1H)-quinazolinones and 3, 4-dihydro-1H-quinazolin-2-thiones.
Sun Q, et al.
Tetrahedron Letters, 42(25), 4119-4121 (2001)
Eric Besson et al.
Langmuir : the ACS journal of surfaces and colloids, 22(20), 8346-8352 (2006-09-20)
This work describes how selective patterning of hydrophobic and hydrophilic areas inside microchannels of microfluidic devices can be achieved by combining well-known chemical protocols and standard photolithography equipment (365 nm). Two techniques have been performed and compared. The first technique
Syntheses of nucleosides designed for combinatorial DNA sequencing.
Welch MB, et al.
Chemistry (Weinheim An Der Bergstrasse, Germany), 5, 951-960 (1999)
Aaron D Mills et al.
Journal of combinatorial chemistry, 9(1), 171-177 (2007-01-09)
A library of 200 2-alkyl-3-alkyloxy-2H-indazole-6-carboxamides was synthesized using parallel solution-phase methods. The indazole cyclization reaction was optimized for library production with the best yields resulting from controlled alcohol/water solvent ratios. The key step, a heterocyclization reaction, proceeds by N,N-bond formation
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service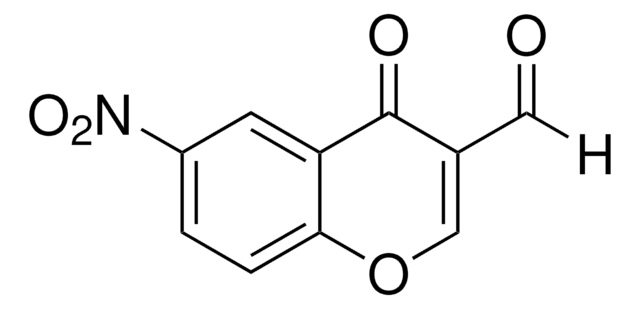
![4-[4-(1-Hydroxyethyl)-2-methoxy-5-nitrophenoxy]butyric acid ≥98.0% (HPLC)](/deepweb/assets/sigmaaldrich/product/structures/232/152/e26ea38f-f1d4-4f88-a61b-466cd10aa1dc/640/e26ea38f-f1d4-4f88-a61b-466cd10aa1dc.png)








