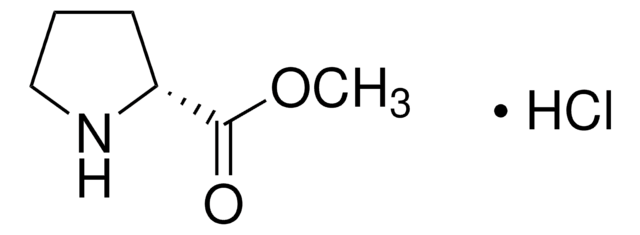
287067
L-Proline methyl ester hydrochloride
98%, for peptide synthesis
Synonym(s):
(S)-Methyl pyrrolidine-2-carboxylate hydrochloride, L-Pro-OMe HCl
About This Item
Recommended Products
product name
L-Proline methyl ester hydrochloride, 98%
Quality Level
Assay
98%
form
solid
optical activity
[α]20/D −31°, c = 0.5 in H2O
reaction suitability
reaction type: solution phase peptide synthesis
mp
69-71 °C (lit.)
application(s)
peptide synthesis
SMILES string
Cl[H].[H][C@]1(CCCN1)C(=O)OC
InChI
1S/C6H11NO2.ClH/c1-9-6(8)5-3-2-4-7-5;/h5,7H,2-4H2,1H3;1H/t5-;/m0./s1
InChI key
HQEIPVHJHZTMDP-JEDNCBNOSA-N
Looking for similar products? Visit Product Comparison Guide
Application
- A building block in peptide synthesis.
- A catalyst to synthesize 3,4-dihydropyrimidin-2-(1H)-ones via Biginelli condensation reaction with aryl aldehydes, methyl acetoacetate, and urea.
- A reactant to prepare prolylproline.
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service