637513
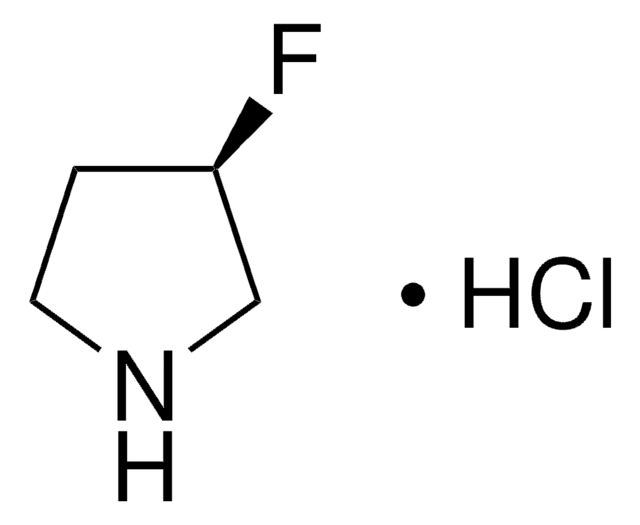
(S)-(+)-3-Fluoropyrrolidine hydrochloride
97%
Synonym(s):
(3S)-(+)-3-Fluoropyrrolidine hydrochloride, (3S)-3-Fluoropyrrolidine hydrochloride
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Empirical Formula (Hill Notation):
C4H8FN · HCl
CAS Number:
Molecular Weight:
125.57
MDL number:
UNSPSC Code:
12352005
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
97%
form
solid
optical activity
[α]20/D +8°, c = 1% in methanol
mp
183-187 °C (lit.)
functional group
fluoro
SMILES string
Cl.F[C@H]1CCNC1
InChI
1S/C4H8FN.ClH/c5-4-1-2-6-3-4;/h4,6H,1-3H2;1H/t4-;/m0./s1
InChI key
LENYOXXELREKGZ-WCCKRBBISA-N
Application
(S)-(+)-3-Fluoropyrrolidine hydrochloride can be employed as a building block for the preparation of:
- Fluorinated pyrrolidine derivatives of cyclohexylglycine amides as potential inhibitors for dipeptidyl peptidase IV.
- Fluoropyrrolidine contaning N--ethylurea pyrazole derivatives as potential and selective inhibitors for Trypanosoma brucei and Trypanosoma cruzi parasitic causative agents.
- Urea derivatives of (S)-2-(3,4-difluorophenyl)-5-(3-fluoro-N-pyrrolidylamido)benzothiazole as potent antitrypanosomal agents.
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 4 Oral - Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Fluoropyrrolidine amides as dipeptidyl peptidase IV inhibitors
Caldwell CG, et al.
Bioorganic & Medicinal Chemistry Letters, 14(5), 1265-1268 (2004)
Urea derivatives of 2-aryl-benzothiazol-5-amines: a new class of potential drugs for human African trypanosomiasis
Patrick DA, et al.
Journal of Medicinal Chemistry, 60(3), 957-971 (2017)
Discovery of Potent N-Ethylurea Pyrazole Derivatives as Dual Inhibitors of Trypanosoma brucei and Trypanosoma cruzi
Varghese S, et al.
ACS Medicinal Chemistry Letters (2019)
Charles G Caldwell et al.
Bioorganic & medicinal chemistry letters, 14(5), 1265-1268 (2004-02-26)
Amides derived from fluorinated pyrrolidines and 4-substituted cyclohexylglycine analogues have been prepared and evaluated as inhibitors of dipeptidyl dipeptidase IV (DP-IV). Analogues which incorporated (S)-3-fluoropyrrolidine showed good selectivity for DP-IV over quiescent cell proline dipeptidase (QPP). Compound 48 had good
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service