85325
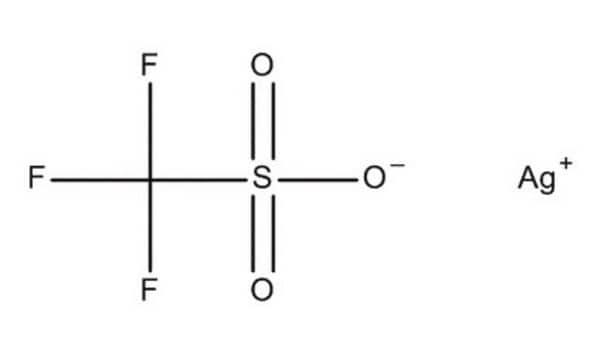
Silver trifluoromethanesulfonate
purum, ≥98.0% (Ag)
Synonym(s):
Ag(OTf), Silver (trifluoromethyl)sulfonate, Silver triflate, Trifluoromethanesulfonic acid silver salt
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Linear Formula:
CF3SO3Ag
CAS Number:
Molecular Weight:
256.94
Beilstein:
3598402
EC Number:
MDL number:
UNSPSC Code:
12161600
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
grade
purum
Quality Level
Assay
≥98.0% (Ag)
form
crystals
reaction suitability
core: silver
reagent type: catalyst
SMILES string
[Ag+].[O-]S(=O)(=O)C(F)(F)F
InChI
1S/CHF3O3S.Ag/c2-1(3,4)8(5,6)7;/h(H,5,6,7);/q;+1/p-1
InChI key
QRUBYZBWAOOHSV-UHFFFAOYSA-M
Related Categories
General description
Silver trifluoromethanesulfonate p-complexes of monoenes, dienes, trienes, monoynes and diynes have been prepared. It reacts with 2-fluoro- and 3-fluoro-4-alkoxystilbazoles to afford the mesomorphic complexes. Iodine monochloride/AgOTf constitutes an efficient promoter system for the O-glycoside synthesis.
Application
Silver trifluoromethanesulfonate (AgOTf) may be employed as a reagent during glucosylation of several alcohols. AgOTf in combination with p-nitrobenzenesulfenyl chloride may be employed as an activator for the glycosylation.
It may be used for the synthesis of the following:
It may be used for the synthesis of the following:
- cystine-containing peptides
- 3-aminoalkylated indoles
- benzo[b]oxepines and 2H-chromenes
- diversely substituted iminoimidazoazines
Other Notes
Reagent for the substitution of halides by triflate.; Reagent for the glycosylation of glycosyl halides; Reagent used for the deprotection of protected thiols
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Dam. 1 - Skin Irrit. 2
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
P.J. Garegg et al.
Acta Chemica Scandinavica. Series B, 33, 116-116 (1979)
V. Pozsgay et al.
The Journal of Organic Chemistry, 46, 3761-3761 (1981)
David Crich et al.
Carbohydrate research, 343(10-11), 1858-1862 (2008-04-01)
p-Nitrobenzenesulfenyl chloride is a stable commercially available sulfenyl chloride that, in conjunction with silver triflate, cleanly activates a wide range of thioglycosides for glycosylation at -78 degrees C in CH(2)Cl(2).
H. Tamamura et al.
Tetrahedron Letters, 34, 4931-4931 (1993)
Silver Triflate-Catalyzed Cyclization of 2-Amino-6-propargyl-amineazines Leading to Iminoimidazoazines.
Chioua M, et al.
Advanced Synthesis & Catalysis, 356(6), 1235-1241 (2014)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service