About This Item
Recommended Products
Assay
≥99%
form
powder
mp
348-350 °C
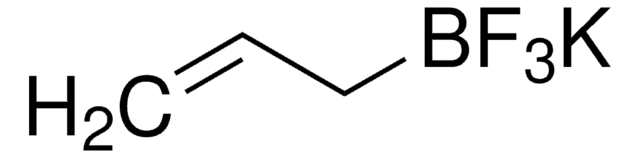
SMILES string
[K+].F[B-](F)(F)C1CC1
InChI
1S/C3H5BF3.K/c5-4(6,7)3-1-2-3;/h3H,1-2H2;/q-1;+1
InChI key
CFMLURFHOSOXRC-UHFFFAOYSA-N
General description
Application
Organotrifluoroborates as versatile and stable boronic acid surrogates.
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Repr. 1B
Storage Class Code
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
Bench-stable Potassium Organotrifluoroborates enable diverse C-C bond formation reactions.
Bench-stable Potassium Organotrifluoroborates enable diverse C-C bond formation reactions.
Bench-stable Potassium Organotrifluoroborates enable diverse C-C bond formation reactions.
Bench-stable Potassium Organotrifluoroborates enable diverse C-C bond formation reactions.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service