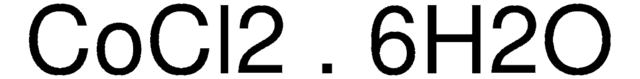223387
Nickel(II) chloride hexahydrate
ReagentPlus®
About This Item
Produits recommandés
Qualité
reagent
Niveau de qualité
Gamme de produits
ReagentPlus®
Forme
powder or crystals
Concentration
23.0-26.0% Ni (EDTA titration)
Chaîne SMILES
[H]O[H].[H]O[H].[H]O[H].[H]O[H].[H]O[H].[H]O[H].Cl[Ni]Cl
InChI
1S/2ClH.Ni.6H2O/h2*1H;;6*1H2/q;;+2;;;;;;/p-2
Clé InChI
LAIZPRYFQUWUBN-UHFFFAOYSA-L
Vous recherchez des produits similaires ? Visite Guide de comparaison des produits
Description générale
Application
- Synthesis of tetra-substituted pyrrole derivatives via four-component condensation reaction of aromatic aldehydes, benzylamines, β-ketoesters and nitroalkanes.
- In combination with 1,2-ethanedithiol for the deprotection of tetrahydropyranyl (THP) and tert-butyldimethylsilyl (TBS) ethers to their corresponding hydroxyl compounds.
- Synthesis of 3,4-dihydropyrimidinones from aldehydes and β-keto esters via Biginelli condensation.
Informations légales
Mention d'avertissement
Danger
Mentions de danger
Conseils de prudence
Classification des risques
Acute Tox. 3 Inhalation - Acute Tox. 3 Oral - Aquatic Acute 1 - Aquatic Chronic 1 - Carc. 1A Inhalation - Muta. 2 - Repr. 1B - Resp. Sens. 1 - Skin Irrit. 2 - Skin Sens. 1 - STOT RE 1 Inhalation
Organes cibles
Lungs
Code de la classe de stockage
6.1D - Non-combustible acute toxic Cat.3 / toxic hazardous materials or hazardous materials causing chronic effects
Classe de danger pour l'eau (WGK)
WGK 3
Point d'éclair (°F)
Not applicable
Point d'éclair (°C)
Not applicable
Faites votre choix parmi les versions les plus récentes :
Déjà en possession de ce produit ?
Retrouvez la documentation relative aux produits que vous avez récemment achetés dans la Bibliothèque de documents.
Les clients ont également consulté
Articles
Nickel complexes catalyze various synthetic reactions like oxidative addition, C-H activation, and cross-coupling.
Nickel complexes catalyze various synthetic reactions like oxidative addition, C-H activation, and cross-coupling.
Nickel complexes catalyze various synthetic reactions like oxidative addition, C-H activation, and cross-coupling.
Nickel complexes catalyze various synthetic reactions like oxidative addition, C-H activation, and cross-coupling.
Notre équipe de scientifiques dispose d'une expérience dans tous les secteurs de la recherche, notamment en sciences de la vie, science des matériaux, synthèse chimique, chromatographie, analyse et dans de nombreux autres domaines..
Contacter notre Service technique