T4062
Tetracycline hydrochloride
meets USP testing specifications
Synonym(s):
Tetracycline HCL
About This Item
Recommended Products
Agency
USP/NF
meets USP testing specifications
Quality Level
Assay
≥900 (Units in µg/mg)
form
crystalline powder
optical activity
[α]/D -255 to 240° (Specific rotation )
storage condition
(Keep container tightly closed in a dry and well-ventilated place. Light sensitive.)
impurities
≤4.0%
color
yellow
mp
220-223 °C (lit.)
antibiotic activity spectrum
Gram-negative bacteria
Gram-positive bacteria
application(s)
pharmaceutical (small molecule)
Mode of action
protein synthesis | interferes
storage temp.
−20°C
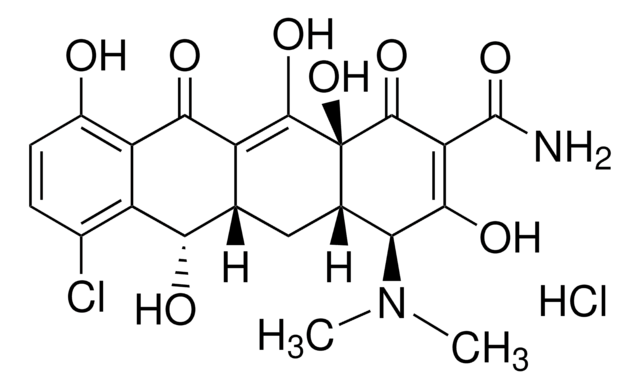
SMILES string
Cl.CN(C)[C@H]1[C@@H]2C[C@H]3C(=C(O)[C@]2(O)C(=O)C(C(N)=O)=C1O)C(=O)c4c(O)cccc4[C@@]3(C)O
InChI
1S/C22H24N2O8.ClH/c1-21(31)8-5-4-6-11(25)12(8)16(26)13-9(21)7-10-15(24(2)3)17(27)14(20(23)30)19(29)22(10,32)18(13)28;/h4-6,9-10,15,25,27-28,31-32H,7H2,1-3H3,(H2,23,30);1H/t9-,10-,15-,21+,22-;/m0./s1
InChI key
XMEVHPAGJVLHIG-FMZCEJRJSA-N
Looking for similar products? Visit Product Comparison Guide
General description
Application
Biochem/physiol Actions
Mode of Resistance: The effects are inactivated via a loss of cell wall permeability.
Antimicrobial spectrum: Includes a wide range of antimicrobial activity against gram-positive and gram-negative bacteria.
Caution
Preparation Note
Other Notes
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Aquatic Acute 1 - Aquatic Chronic 2 - Eye Irrit. 2 - Repr. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 2
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
Choose from one of the most recent versions:
Certificates of Analysis (COA)
Don't see the Right Version?
If you require a particular version, you can look up a specific certificate by the Lot or Batch number.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service