W259500
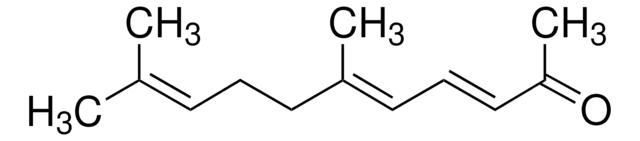
β-Ionone
predominantly trans, ≥97%, FCC, FG
Synonym(s):
4-(2,6,6-Trimethyl-1-cyclohexenyl)-3-buten-2-one
About This Item
Recommended Products
biological source
synthetic
Quality Level
grade
FG
Fragrance grade
Kosher
Agency
follows IFRA guidelines
reg. compliance
EU Regulation 1223/2009
EU Regulation 1334/2008 & 178/2002
FCC
FDA 21 CFR 117
FDA 21 CFR 172.515
Assay
≥97%
refractive index
n20/D 1.52 (lit.)
bp
126-128 °C/12 mmHg (lit.)
density
0.945 g/mL at 25 °C (lit.)
application(s)
flavors and fragrances
Documentation
see Safety & Documentation for available documents
food allergen
no known allergens
fragrance allergen
no known allergens
Organoleptic
berry; woody; floral; sweet
SMILES string
CC(=O)\C=C\C1=C(C)CCCC1(C)C
InChI
1S/C13H20O/c1-10-6-5-9-13(3,4)12(10)8-7-11(2)14/h7-8H,5-6,9H2,1-4H3/b8-7+
InChI key
PSQYTAPXSHCGMF-BQYQJAHWSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
General description
Biochem/physiol Actions
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Aquatic Chronic 2 - Skin Irrit. 2
Storage Class Code
10 - Combustible liquids
WGK
WGK 2
Personal Protective Equipment
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service






![1,3,4,6,7,8-Hexahydro-4,6,6,7,8,8-hexamethylcyclopenta[g]-2-benzopyran certified reference material, TraceCERT®, mixture of isomers, Manufactured by: Sigma-Aldrich Production GmbH, Switzerland](/deepweb/assets/sigmaaldrich/product/structures/388/630/32c6a671-64a6-4e33-9e15-ec3e0a11ff3d/640/32c6a671-64a6-4e33-9e15-ec3e0a11ff3d.png)