45199
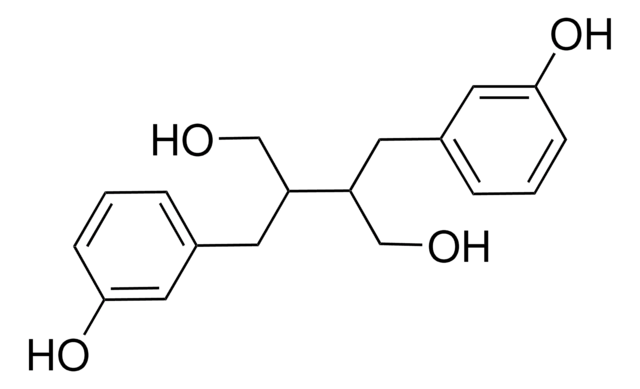
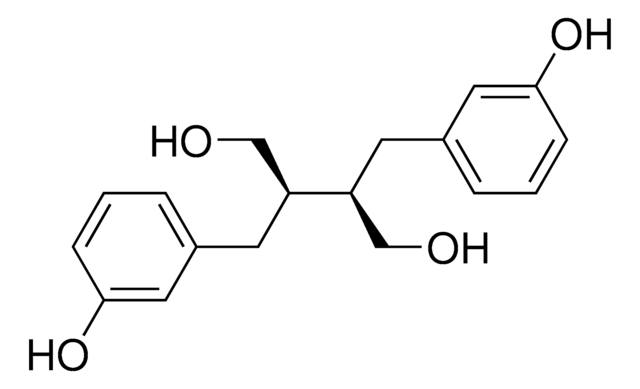
Enterolactone
~95% (HPLC)
Synonym(s):
trans-α,β-Bis(3-hydroxybenzyl)butyrolactone
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Empirical Formula (Hill Notation):
C18H18O4
CAS Number:
Molecular Weight:
298.33
Beilstein:
4699604
MDL number:
UNSPSC Code:
85151701
PubChem Substance ID:
NACRES:
NA.77
Recommended Products
Assay
~95% (HPLC)
form
solid
storage temp.
2-8°C
SMILES string
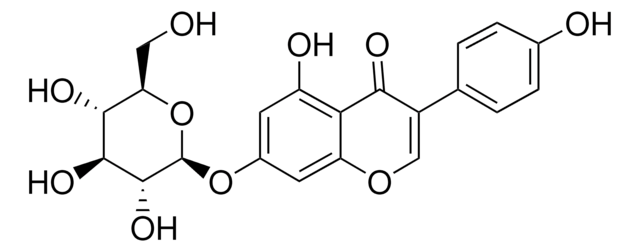
Oc1cccc(C[C@@H]2COC(=O)[C@H]2Cc3cccc(O)c3)c1
InChI
1S/C18H18O4/c19-15-5-1-3-12(8-15)7-14-11-22-18(21)17(14)10-13-4-2-6-16(20)9-13/h1-6,8-9,14,17,19-20H,7,10-11H2/t14-,17+/m1/s1
InChI key
HVDGDHBAMCBBLR-PBHICJAKSA-N
Looking for similar products? Visit Product Comparison Guide
Biochem/physiol Actions
Enterolactone is the major human metabolite of dietary sesamin (lignan from sesame seeds). A phytoestrogen, it shows an inverse correlation with risk of breast cancer in one study.
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Mark J McCann et al.
Molecular nutrition & food research, 57(2), 212-224 (2012-11-14)
There is evidence that a mammalian lignan, enterolactone (ENL), decreases the proliferation rate of prostate cancer cells, although previous studies have used concentrations difficult to achieve through dietary modification. We have therefore investigated the anti-proliferative effects of ENL in an
Anne Tuomisto et al.
Scientific reports, 9(1), 11209-11209 (2019-08-03)
The dietary lignan metabolite, enterolactone, has been suggested to have anti-cancer functions, and high serum enterolactone concentrations have been associated with decreased risk of breast and prostate cancers. We hypothesized that serum enterolactone concentrations as a marker of plant-based foods
Li-Quan Wang
Journal of chromatography. B, Analytical technologies in the biomedical and life sciences, 777(1-2), 289-309 (2002-09-25)
The mammalian phytoestrogens enterodiol (END) and enterolactone (ENL) are produced in the colon by the action of bacteria on the plant precursors matairesinol (MAT), secoisolariciresinol (SECO), their glycosides, and other precursors in the diet. Both END and ENL have been
Julie Aarestrup et al.
The British journal of nutrition, 109(12), 2269-2275 (2012-11-02)
The phyto-oestrogen enterolactone has been hypothesised to protect against hormone-dependent cancers, probably through its antioestrogenic potential. We investigated whether a higher level of plasma enterolactone was associated with a lower incidence of endometrial cancer in a case-cohort study in the
Yulan Lin et al.
The British journal of nutrition, 109(10), 1873-1880 (2012-09-26)
The validity of using FFQ to assess dietary lignans is uncertain. We aimed to validate the use of FFQ for the assessment of dietary intake of lignans compared to the serum biomarker enterolactone, the main product of dietary lignans' metabolism
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service