All Photos(1)
About This Item
Empirical Formula (Hill Notation):
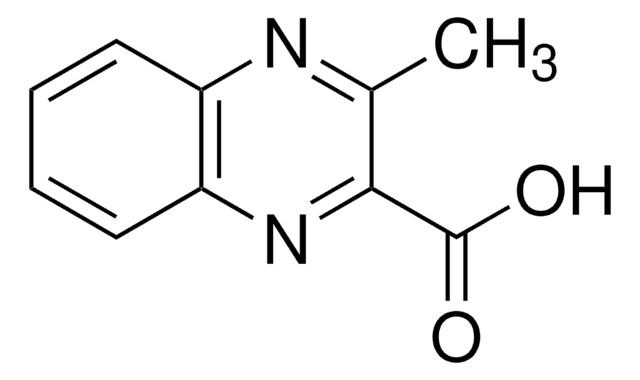
C9H6N2O2
CAS Number:
Molecular Weight:
174.16
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
97%
mp
208 °C (dec.) (lit.)
functional group
carboxylic acid
SMILES string
OC(=O)c1cnc2ccccc2n1
InChI
1S/C9H6N2O2/c12-9(13)8-5-10-6-3-1-2-4-7(6)11-8/h1-5H,(H,12,13)
InChI key
UPUZGXILYFKSGE-UHFFFAOYSA-N
General description
Linear and Freundlich adsorption isotherm coefficient of 2-quinoxalinecarboxylic acid has been evaluated.
Application
2-Quinoxalinecarboxylic acid has been used in the preparation of:
- N-(2-quinoxaloyl)-α-amino acids
- bisquinoxaloyl (bisquinoxalinecarbonyl) derivatives
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Certificates of Analysis (COA)
Lot/Batch Number
Don't see the Right Version?
If you require a particular version, you can look up a specific certificate by the Lot or Batch number.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
N Prabavathi et al.
Spectrochimica acta. Part A, Molecular and biomolecular spectroscopy, 92, 325-335 (2012-03-27)
The FTIR and FT-Raman spectra of 2-quinoxaline carboxylic acid (2-QCA) has been recorded in the region 4000-450 and 4000-100 cm(-1), respectively. The conformational analysis, optimized geometry, frequency and intensity of the vibrational bands of 2-QCA were obtained by the density
M D Rose et al.
Food additives and contaminants, 12(2), 177-183 (1995-03-01)
A method for the determination of residues of quinoxaline-2-carboxylic acid (QCA), the major metabolite of carbadox, in pig kidney has been developed. Tissue samples were subjected to alkaline hydrolysis, liquid-liquid extractions, ion-exchange chromatography and further extraction to concentrate the analyte
D Peng et al.
Food additives & contaminants. Part A, Chemistry, analysis, control, exposure & risk assessment, 28(11), 1524-1533 (2011-08-11)
The feed drug additive carbadox is a suspected carcinogen and mutagen. To monitor effectively residues of carbadox in the edible tissues of food-producing animals, an indirect competitive enzyme-linked immunosorbent assay (ic-ELISA) to detect quinoxaline-2-carboxylic acid, the marker residue of carbadox
Lingli Huang et al.
Journal of chromatography. B, Analytical technologies in the biomedical and life sciences, 874(1-2), 7-14 (2008-10-08)
Cyadox (CYX) is an antimicrobial growth-promoter of the quinoxalines. It is highly effective on improving growth and feed conversion of chicken with little toxicity. For food safety concerns, HPLC-UV methods were developed for the sequential determination of CYX and its
Joshua A Hagen et al.
Sensors (Basel, Switzerland), 11(7), 6645-6655 (2011-12-14)
Zinc oxide field effect transistors (ZnO-FET), covalently functionalized with single stranded DNA aptamers, provide a highly selective platform for label-free small molecule sensing. The nanostructured surface morphology of ZnO provides high sensitivity and room temperature deposition allows for a wide
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service