Scegli un formato
| A voi/SKU | Disponibilità | Prezzo |
|---|---|---|
25 mg | Per conoscere la disponibilità, visualizza il carrello | CHF 435.00 |
Informazioni su questo articolo
CHF 435.00
Quality Segment
assay
≥98% (HPLC)
form
powder
composition
Peptide content, ≥85%
solubility
water: 50 mg/mL, clear, colorless
storage temp.
−20°C
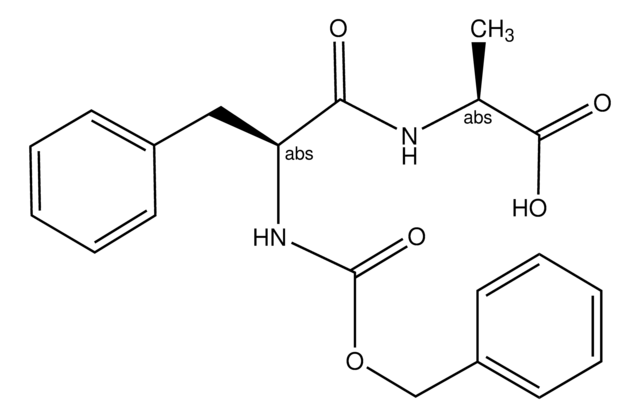
SMILES string
OC([C@@H](C)NC([C@@H](C)NC([C@@H](NC(C)=O)CCCCNC(C)=O)=O)=O)=O
InChI
1S/C16H28N4O6/c1-9(14(23)19-10(2)16(25)26)18-15(24)13(20-12(4)22)7-5-6-8-17-11(3)21/h9-10,13H,5-8H2,1-4H3,(H,17,21)(H,18,24)(H,19,23)(H,20,22)(H,25,26)
InChI key
VIHGYLJIMMKSBR-UHFFFAOYSA-N
General description
1 of 1
Questo articolo | |||
|---|---|---|---|
| assay ≥98% (HPLC) | assay ≥99% (TLC) | assay ≥98% (HPLC) | assay ≥98% (TLC) |
| solubility water: 50 mg/mL, clear, colorless | solubility methanol: ~50 mg/mL, almost clear, almost colorless | solubility water: 50 mg/mL, clear, light yellow | solubility water: 50 mg/mL, clear, colorless to yellow |
| Quality Level 200 | Quality Level 200 | Quality Level 200 | Quality Level 200 |
| form powder | form powder | form powder | form powder |
| storage temp. −20°C | storage temp. −20°C | storage temp. −20°C | storage temp. −20°C |
| composition Peptide content, ≥85% | composition - | composition - | composition - |
Classe di stoccaggio
11 - Combustible Solids
wgk
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Gloves, type N95 (US)
Scegli una delle versioni più recenti:
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.