Scegli un formato
| A voi/SKU | Disponibilità | Prezzo |
|---|---|---|
100 mg | Per conoscere la disponibilità, visualizza il carrello | CHF 95.30 |
250 mg | Per conoscere la disponibilità, visualizza il carrello | CHF 202.00 |
1 g | Per conoscere la disponibilità, visualizza il carrello | CHF 605.00 |
Informazioni su questo articolo
Quality Segment
assay
≥98%
form
powder
drug control
regulated under CDSA - not available from Sigma-Aldrich Canada
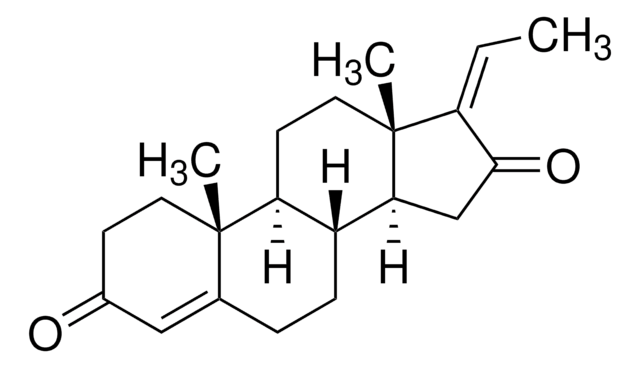
SMILES string
CC12Cc3cnoc3C=C1CCC4C2CCC5(C)C4CC[C@@]5(O)C#C
InChI
1S/C22H27NO2/c1-4-22(24)10-8-18-16-6-5-15-11-19-14(13-23-25-19)12-20(15,2)17(16)7-9-21(18,22)3/h1,11,13,16-18,24H,5-10,12H2,2-3H3/t16?,17?,18?,20?,21?,22-/m0/s1
InChI key
POZRVZJJTULAOH-ZIKCMSSPSA-N
Biochem/physiol Actions
Features and Benefits
Packaging
1 of 1
Questo articolo | |||
|---|---|---|---|
| form powder | form - | form powder | form powder |
| assay ≥98% | assay - | assay ≥98% (HPLC) | assay ≥89% (HPLC) |
| drug control regulated under CDSA - not available from Sigma-Aldrich Canada | drug control regulated under CDSA - not available from Sigma-Aldrich Canada | drug control - | drug control - |
| Quality Level 200 | Quality Level - | Quality Level - | Quality Level 200 |
Still not finding the right product?
Explore all of our products under Danazol
signalword
Warning
hcodes
Hazard Classifications
Acute Tox. 4 Dermal - Acute Tox. 4 Inhalation - Repr. 2
Classe di stoccaggio
11 - Combustible Solids
wgk
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Gloves, type P3 (EN 143) respirator cartridges
Scegli una delle versioni più recenti:
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.