Scegli un formato
| A voi/SKU | Disponibilità | Prezzo |
|---|---|---|
25 mg | Per conoscere la disponibilità, visualizza il carrello | CHF 145.00 |
Informazioni su questo articolo
Nome del prodotto
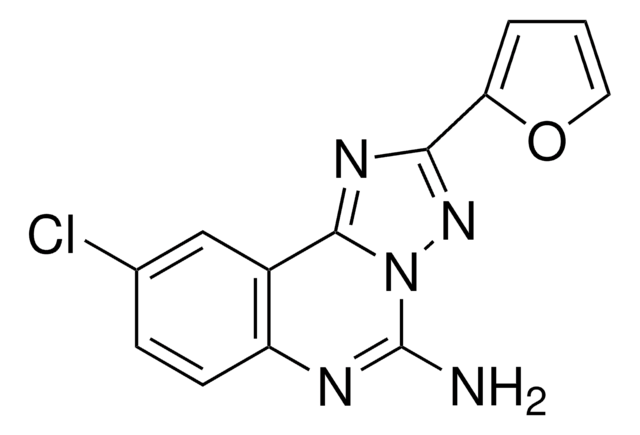
CGS-15943, solid
assay
≥98% (HPLC)
Quality Segment
form
solid
color
white
solubility
DMSO: >10 mg/mL, H2O: insoluble
storage temp.
room temp
SMILES string
Nc1nc2ccc(Cl)cc2c3nc(nn13)-c4ccco4
InChI
1S/C13H8ClN5O/c14-7-3-4-9-8(6-7)12-17-11(10-2-1-5-20-10)18-19(12)13(15)16-9/h1-6H,(H2,15,16)
InChI key
MSJODEOZODDVGW-UHFFFAOYSA-N
Gene Information
human ... ADORA1(134), ADORA2A(135), ADORA2B(136), ADORA3(140)
rat ... Adora1(29290), Adora2a(25369), Adora2b(29316)
Application
Biochem/physiol Actions
1 of 1
Questo articolo | |||
|---|---|---|---|
| form solid | form solid | form powder | form powder |
| assay ≥98% (HPLC) | assay ≥98% (HPLC) | assay ≥98% (HPLC) | assay ≥98% (HPLC) |
| Quality Level 100 | Quality Level 100 | Quality Level 100 | Quality Level 100 |
| storage temp. room temp | storage temp. - | storage temp. 2-8°C | storage temp. room temp |
| solubility DMSO: >10 mg/mL, H2O: insoluble | solubility DMSO: soluble >10 mg/mL, H2O: insoluble | solubility DMSO: ≥20 mg/mL | solubility DMSO: >15 mg/mL |
| color white | color off-white | color light tan to tan | color white to tan |
ppe
Eyeshields, Gloves, type N95 (US)
Classe di stoccaggio
11 - Combustible Solids
wgk
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
Scegli una delle versioni più recenti:
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.