392529
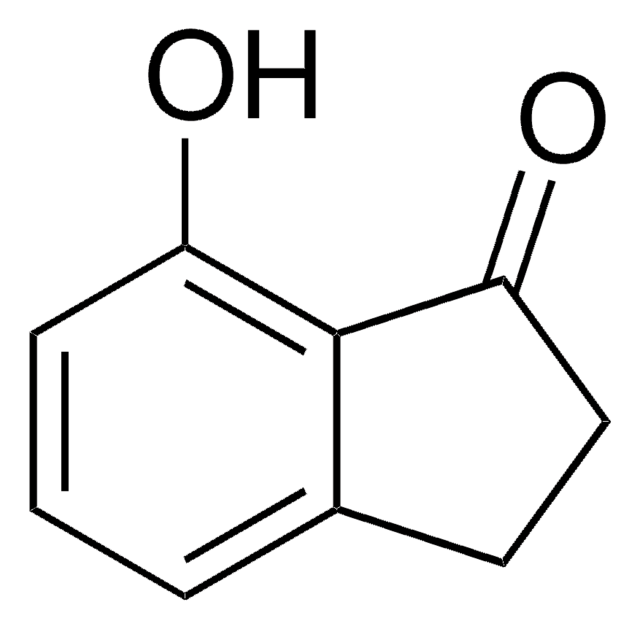
4-Methoxy-1-indanone
99%
Autenticatiper visualizzare i prezzi riservati alla tua organizzazione & contrattuali
About This Item
Formula empirica (notazione di Hill):
C10H10O2
Numero CAS:
Peso molecolare:
162.19
Numero MDL:
Codice UNSPSC:
12352100
ID PubChem:
NACRES:
NA.22
Prodotti consigliati
Livello qualitativo
Saggio
99%
P. eboll.
115-120 °C/0.5 mmHg (lit.)
Punto di fusione
105-107 °C (dec.) (lit.)
Stringa SMILE
COc1cccc2C(=O)CCc12
InChI
1S/C10H10O2/c1-12-10-4-2-3-7-8(10)5-6-9(7)11/h2-4H,5-6H2,1H3
BTYSYELHQDGJAB-UHFFFAOYSA-N
Descrizione generale
4-Methoxy-1-indanone, a benzo-fused ketone is a 1-indanone derivative. Its synthesis has been reported.
Applicazioni
4-Methoxy-1-indanone is suitable for use in the comparative study of the effect of different substituents on various benzo-fused ketones on the reaction kinetics by studying the biocatalysed oxidation reaction.
It may be used in the following studies:
It may be used in the following studies:
- Synthesis of isomeric mixture of oximes.
- As a starting material in the synthesis of benzo-fused indolizidine and 4-methoxy-1-indanyl compound.
- Synthesis of 4-methoxy-5-nitro-1-indanone by nitration reaction.
- As one of the reactant in the synthesis of E-2-chloro-8-methyl-3-[(4′-methoxy-1′-indanoyl)-2′-methyliden]-quinoline (IQ), a quinoline derivative.
Codice della classe di stoccaggio
11 - Combustible Solids
Classe di pericolosità dell'acqua (WGK)
WGK 3
Punto d’infiammabilità (°F)
Not applicable
Punto d’infiammabilità (°C)
Not applicable
Dispositivi di protezione individuale
Eyeshields, Gloves, type N95 (US)
Certificati d'analisi (COA)
Cerca il Certificati d'analisi (COA) digitando il numero di lotto/batch corrispondente. I numeri di lotto o di batch sono stampati sull'etichetta dei prodotti dopo la parola ‘Lotto’ o ‘Batch’.
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
W H Pearson et al.
The Journal of organic chemistry, 65(21), 7158-7174 (2000-10-14)
The intramolecular capture of benzocyclobutyl, benzocyclopentyl, and benzocyclohexyl carbocations 7 by azides produces spirocyclic aminodiazonium ions 8, which undergo 1,2-C-to-N rearrangement with loss of dinitrogen to produce benzo-fused iminium ions resulting from either aryl (9) or alkyl (10) migration to
Juan Rodrigues et al.
Memorias do Instituto Oswaldo Cruz, 104(6), 865-870 (2009-10-31)
E-2-chloro-8-methyl-3-[(4'-methoxy-1'-indanoyl)-2'-methyliden]-quinoline (IQ) is a new quinoline derivative which has been reported as a haemoglobin degradation and ss-haematin formation inhibitor. The haemoglobin proteolysis induced by Plasmodium parasites represents a source of amino acids and haeme, leading to oxidative stress in infected
Enzymatic Baeyer-Villiger Oxidation of Benzo-Fused Ketones: Formation of Regiocomplementary Lactones.
Rioz-Martinez A, et al.
European Journal of Organic Chemistry, 2009(15), 2526-2532 (2009)
Beckmann rearrangements of 1-indanone oxime derivatives using aluminum chloride and mechanistic considerations.
Lee BS, et al.
Bull. Korean Chem. Soc., 21(9), 860-866 (2000)
Ju-Ok Lim et al.
European journal of medicinal chemistry, 44(1), 322-331 (2008-04-15)
A series of bicyclic analogues having indan and tetrahydronaphthalene templates in the A-region were designed as conformationally constrained analogues of our previously reported potent TRPV1 antagonists (1, 3). The activities for rat TRPV1 of the conformationally restricted analogues were moderately
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.




![[1,1′-Bis(diphenylphosphino)ferrocene]dichloropalladium(II)](/deepweb/assets/sigmaaldrich/product/structures/130/734/8846aa26-1858-458a-998d-8c306c13bf0f/640/8846aa26-1858-458a-998d-8c306c13bf0f.png)

