122408
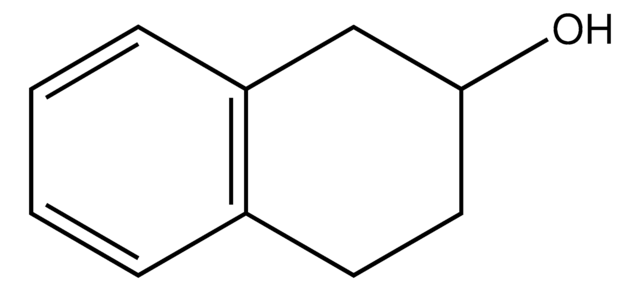
1,2,3,4-Tetrahydro-1-naphthol
97%
Sinonimo/i:
α-Tetralol
Autenticatiper visualizzare i prezzi riservati alla tua organizzazione & contrattuali
About This Item
Formula empirica (notazione di Hill):
C10H12O
Numero CAS:
Peso molecolare:
148.20
Beilstein:
2046227
Numero CE:
Numero MDL:
Codice UNSPSC:
12352100
ID PubChem:
NACRES:
NA.22
Prodotti consigliati
Saggio
97%
Forma fisica
solid
Indice di rifrazione
n20/D 1.564 (lit.)
P. eboll.
102-104 °C/2 mmHg (lit.)
Densità
1.09 g/mL at 25 °C (lit.)
Stringa SMILE
OC1CCCc2ccccc12
InChI
1S/C10H12O/c11-10-7-3-5-8-4-1-2-6-9(8)10/h1-2,4,6,10-11H,3,5,7H2
JAAJQSRLGAYGKZ-UHFFFAOYSA-N
Cerchi prodotti simili? Visita Guida al confronto tra prodotti
Descrizione generale
(R)-(-)-enantiomer of 1,2,3,4-Tetrahydro-1-naphthol is a substrate for aryl sulfotransferase (AST) IV enzyme and (S)-(+)-1,2,3,4-tetrahydro-1-naphthol is a competitive inhibitor of AST IV-catalyzed sulfation of 1-naphthalenemethanol. It is the major urinary metabolite of tetralin.
Applicazioni
1,2,3,4-Tetrahydro-1-naphthol was used as chiral probe to examine the role of three aromatic residues in enzyme-substrate interactions at the sulfuryl acceptor binding site of aryl sulfotransferase IV enzyme.
Avvertenze
Warning
Indicazioni di pericolo
Consigli di prudenza
Classi di pericolo
Acute Tox. 4 Oral
Codice della classe di stoccaggio
11 - Combustible Solids
Classe di pericolosità dell'acqua (WGK)
WGK 3
Dispositivi di protezione individuale
dust mask type N95 (US), Eyeshields, Gloves
Scegli una delle versioni più recenti:
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
Jonathan J Sheng et al.
Drug metabolism and disposition: the biological fate of chemicals, 32(5), 559-565 (2004-04-22)
Aryl sulfotransferase (AST) IV (also named tyrosine-ester sulfotransferase and ST1A1) is a major phenol sulfotransferase in the rat, and it catalyzes the sulfation of many drugs, carcinogens, and other xenobiotics that contain phenol, benzylic alcohol, N-hydroxy arylamine, and oxime functional
Metabolism of tetralin and toxicity of Cuprex in man.
D E Drayer et al.
Drug metabolism and disposition: the biological fate of chemicals, 1(3), 577-579 (1973-05-01)
Vyas Sharma et al.
Journal of medicinal chemistry, 45(25), 5514-5522 (2002-12-03)
Comparative Molecular Field Analysis (CoMFA) methods were used to produce a 3D-QSAR model that correlated the catalytic efficiency of rat hepatic aryl sulfotransferase (AST) IV, expressed as log(k(cat)/K(m)), with the molecular structures of its substrates. A total of 35 substrate
W F Leebaw et al.
The Journal of clinical endocrinology and metabolism, 47(3), 480-487 (1978-09-01)
Although the role of the neurotransmitter, dopamine (DA), in the regulation of PRL has been well documented, controversy exists regarding its participation in the regulation of the other pituitary hormones. Consequently, we infused DA into six healthy male subjects (ages
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.
![[Pd(OAc)2]3 reagent grade, 98%](/deepweb/assets/sigmaaldrich/product/structures/508/249/99a0ef2c-b77c-4d73-8ed9-0cca05b6b41f/640/99a0ef2c-b77c-4d73-8ed9-0cca05b6b41f.png)