F2645
Anti-Factor IX antibody, Mouse monoclonal
clone HIX-1, purified from hybridma cell culture
Synonyme(s) :
Monoclonal Anti-Factor IX
About This Item
Produits recommandés
Source biologique
mouse
Conjugué
unconjugated
Forme d'anticorps
purified immunoglobulin
Type de produit anticorps
primary antibodies
Clone
HIX-1, monoclonal
Forme
buffered aqueous solution
Espèces réactives
human
Technique(s)
indirect ELISA: suitable
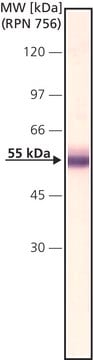
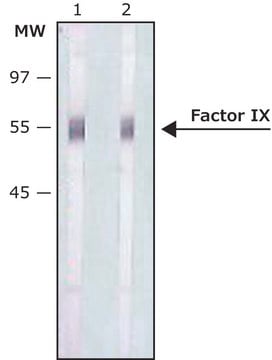
western blot: 2-4 μg/mL
Isotype
IgG1
Numéro d'accès UniProt
Conditions d'expédition
dry ice
Température de stockage
−20°C
Modification post-traductionnelle de la cible
unmodified
Informations sur le gène
human ... F9(2158)
Description générale
Spécificité
This antibody may be used for purification of Factor IX and preparation of Factor IX depleted human plasma.
Immunogène
Application
Enzyme-linked immunosorbent assay (1 paper)
Actions biochimiques/physiologiques
Forme physique
Clause de non-responsabilité
Not finding the right product?
Try our Outil de sélection de produits.
En option
Code de la classe de stockage
12 - Non Combustible Liquids
Classe de danger pour l'eau (WGK)
nwg
Point d'éclair (°F)
Not applicable
Point d'éclair (°C)
Not applicable
Certificats d'analyse (COA)
Recherchez un Certificats d'analyse (COA) en saisissant le numéro de lot du produit. Les numéros de lot figurent sur l'étiquette du produit après les mots "Lot" ou "Batch".
Déjà en possession de ce produit ?
Retrouvez la documentation relative aux produits que vous avez récemment achetés dans la Bibliothèque de documents.
Notre équipe de scientifiques dispose d'une expérience dans tous les secteurs de la recherche, notamment en sciences de la vie, science des matériaux, synthèse chimique, chromatographie, analyse et dans de nombreux autres domaines..
Contacter notre Service technique