Sélectionner une taille de conditionnement
| À vous/Référence | Disponibilité | Prix |
|---|---|---|
100 mg | Vérifier la disponibilité des articles du panier | 85.10 CHF |
500 mg | Vérifier la disponibilité des articles du panier | 310.00 CHF |
A propos de cet article
product line
BioReagent
Quality Segment
assay
~90% (HPLC)
form
powder
solubility
DMF: soluble, DMSO: soluble, aqueous base: soluble
fluorescence
λex 350 nm; λem 433 nm in methanol
suitability
suitable for fluorescence
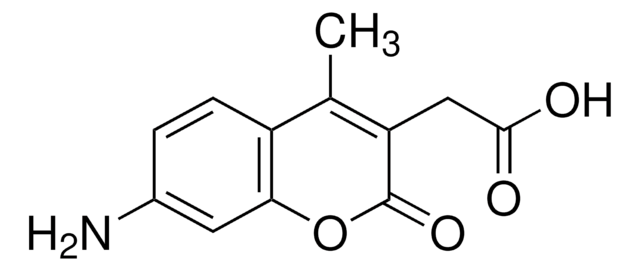
InChI
1S/C12H11NO4/c1-6-8-3-2-7(13)4-10(8)17-12(16)9(6)5-11(14)15/h2-4H,5,13H2,1H3,(H,14,15)
InChI key
QEQDLKUMPUDNPG-UHFFFAOYSA-N
Application
Packaging
Other Notes
1 of 1
Cet article | |||
|---|---|---|---|
| suitability suitable for fluorescence | suitability - | suitability - | suitability - |
| fluorescence λex 350 nm; λem 433 nm in methanol | fluorescence - | fluorescence - | fluorescence - |
| solubility DMF: soluble, aqueous base: soluble, DMSO: soluble | solubility DMF: soluble 50 mg/mL, clear, colorless to yellow | solubility - | solubility - |
| form powder | form - | form liquid | form liquid |
| assay ~90% (HPLC) | assay 97% | assay ≥99.8% (acidimetric) | assay ≥99.8% (alkalimetric) |
| Quality Level 100 | Quality Level 200 | Quality Level 100 | Quality Level 100 |
signalword
Warning
hcodes
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
target_organs
Respiratory system
Classe de stockage
11 - Combustible Solids
wgk
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
dust mask type N95 (US), Eyeshields, Gloves
Faites votre choix parmi les versions les plus récentes :
Déjà en possession de ce produit ?
Retrouvez la documentation relative aux produits que vous avez récemment achetés dans la Bibliothèque de documents.