8.52017
Fmoc-Pro-OH
Novabiochem®
Synonyme(s) :
Fmoc-Pro-OH, N-α-Fmoc-L-proline
About This Item
Produits recommandés
Niveau de qualité
Gamme de produits
Novabiochem®
Pureté
≥94.0% (acidimetric)
≥98% (TLC)
≥99.0% (HPLC)
Forme
powder
Capacité de réaction
reaction type: Fmoc solid-phase peptide synthesis
Fabricant/nom de marque
Novabiochem®
Pf
112-115 °C
Application(s)
peptide synthesis
Groupe fonctionnel
Fmoc
Température de stockage
2-30°C
InChI
1S/C20H19NO4/c22-19(23)18-10-5-11-21(18)20(24)25-12-17-15-8-3-1-6-13(15)14-7-2-4-9-16(14)17/h1-4,6-9,17-18H,5,10-12H2,(H,22,23)
Clé InChI
ZPGDWQNBZYOZTI-UHFFFAOYSA-N
Description générale
Standard building block for introduction of proline amino-acid residues by Fmoc SPPS
Associated Protocols and Technical Articles
Fmoc-amino acids for Peptide Production
Cleavage and Deprotection Protocols for Fmoc SPPS
Application
- Biphasic electrochemical peptide synthesis: This study highlights the application of Fmoc-Pro-OH in electrochemical peptide synthesis, where slight excesses of reagents and electricity were required, possibly due to steric hindrance (S Nagahara, Y Okada, Y Kitano, K Chiba - Chemical Science, 2021).
- Elucidation of the Mechanism of Endo-XaaC-terminal Peptide Impurity Formation in SPPS: Fmoc-Pro-OH′s role was analyzed in a study investigating impurity formation during solid-phase peptide synthesis, highlighting its distinctively low tendency to form impurities (Y Yang, L Hansen, A Baldi - Organic Process Research & Development, 2021).
- Polymer–peptide delivery platforms: This research utilized Fmoc-Pro-OH in the development of polymer-based DNA delivery systems, emphasizing the effect of oligopeptide orientation on delivery efficacy (SS Parelkar, R Letteri, D Chan-Seng - Biomacromolecules, 2014).
- Structure–Activity Relationship Study of N-Hydroxyphtalimide Derivatives: The study explored the use of Fmoc-Pro-OH in the synthesis of N-Hydroxyphtalimide derivatives for detecting amines during peptide synthesis (K Takamatsu, R Suzuki, A Matsunaga - The Journal of Organic Chemistry, 2023).
Liaison
Remarque sur l'analyse
Appearance of substance (visual): powder
Colour index (0,5 M in DMF): ≤ 150 Hazen
Identity (IR): passes test
Enantiomeric purity: ≥ 99.8 % (a/a)
Purity (HPLC): ≥ 99.0 % (a/a)
Fmoc-ß-Ala-OH (HPLC): ≤ 0.1 % (a/a)
Fmoc-β-Ala-Pro-OH (HPLC): ≤ 0.1 % (a/a)
Fmoc-Pro-Pro-OH (HPLC): ≤ 0.1 % (a/a)
Assay free amino acid (GC): ≤ 0.2 %
Purity (TLC(011A)): ≥ 98 %
Solubility (25 mmole in 50 ml DMF): clearly soluble
Assay (acidimetric): ≥ 94.0 %
Water (K. F.): ≤ 6.0 %
Ethyl acetate (HS-GC): ≤ 0.5 %
Acetate (IC): ≤ 0.02 %
To see the solvent systems used for TLC of Novabiochem® products please click here.
Informations légales
Not finding the right product?
Try our Outil de sélection de produits.
Code de la classe de stockage
11 - Combustible Solids
Classe de danger pour l'eau (WGK)
WGK 3
Point d'éclair (°F)
Not applicable
Point d'éclair (°C)
Not applicable
Certificats d'analyse (COA)
Recherchez un Certificats d'analyse (COA) en saisissant le numéro de lot du produit. Les numéros de lot figurent sur l'étiquette du produit après les mots "Lot" ou "Batch".
Déjà en possession de ce produit ?
Retrouvez la documentation relative aux produits que vous avez récemment achetés dans la Bibliothèque de documents.
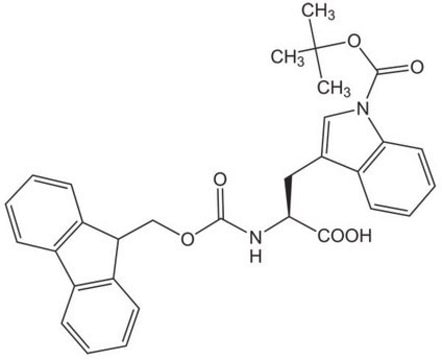
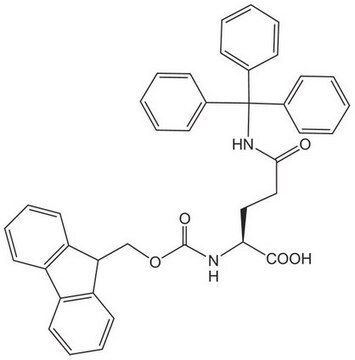
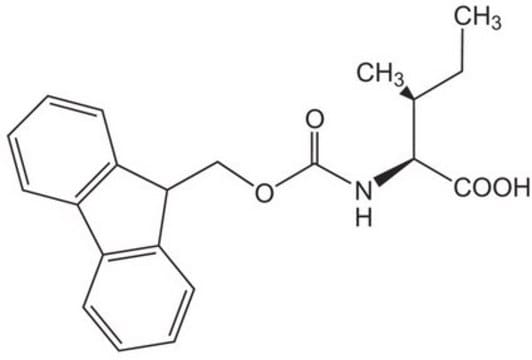
Les clients ont également consulté
Articles
Proline analogues are promising candidates for tuning the biological, pharmaceutical, or physicochemical properties of naturally occuring, as well as de novo designed, linear, and, cyclic peptides.
Contenu apparenté
Purer Fmocs Means Purer Peptides
Notre équipe de scientifiques dispose d'une expérience dans tous les secteurs de la recherche, notamment en sciences de la vie, science des matériaux, synthèse chimique, chromatographie, analyse et dans de nombreux autres domaines..
Contacter notre Service technique