586765
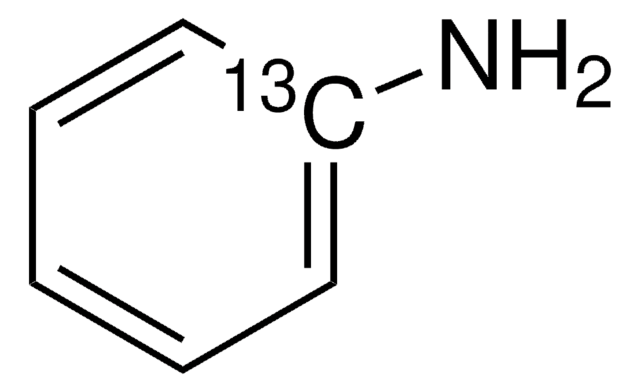
Aniline-4-13C
99 atom % 13C
Synonyme(s) :
Benzenamine-4-13C
About This Item
Produits recommandés
Pureté isotopique
99 atom % 13C
Point d'ébullition
184 °C (lit.)
Pf
-6 °C (lit.)
Densité
1.033 g/mL at 25 °C
Changement de masse
M+1
Température de stockage
2-8°C
Chaîne SMILES
Nc1cc[13cH]cc1
InChI
1S/C6H7N/c7-6-4-2-1-3-5-6/h1-5H,7H2/i1+1
Clé InChI
PAYRUJLWNCNPSJ-OUBTZVSYSA-N
Catégories apparentées
Conditionnement
Mention d'avertissement
Danger
Mentions de danger
Classification des risques
Acute Tox. 3 Dermal - Acute Tox. 3 Inhalation - Acute Tox. 3 Oral - Aquatic Acute 1 - Aquatic Chronic 1 - Carc. 2 - Eye Dam. 1 - Muta. 2 - Resp. Sens. 1
Code de la classe de stockage
6.1A - Combustible acute toxic Cat. 1 and 2 / very toxic hazardous materials
Classe de danger pour l'eau (WGK)
WGK 3
Point d'éclair (°F)
168.8 °F - closed cup
Point d'éclair (°C)
76 °C - closed cup
Certificats d'analyse (COA)
Recherchez un Certificats d'analyse (COA) en saisissant le numéro de lot du produit. Les numéros de lot figurent sur l'étiquette du produit après les mots "Lot" ou "Batch".
Déjà en possession de ce produit ?
Retrouvez la documentation relative aux produits que vous avez récemment achetés dans la Bibliothèque de documents.
Notre équipe de scientifiques dispose d'une expérience dans tous les secteurs de la recherche, notamment en sciences de la vie, science des matériaux, synthèse chimique, chromatographie, analyse et dans de nombreux autres domaines..
Contacter notre Service technique