8.17076
TWEEN® 60 (Polysorbate)
EMPROVE® ESSENTIAL, Ph. Eur., JPE, NF
Fabrication pharma
Synonyme(s) :
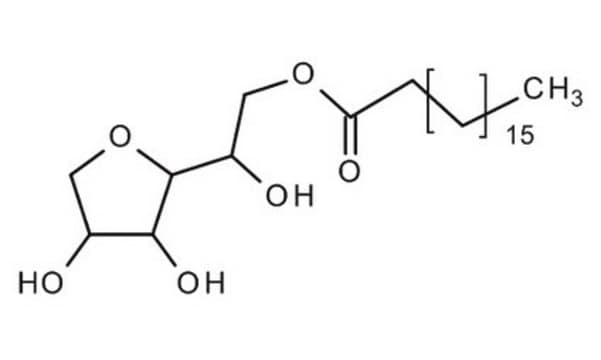
Polyoxyethylene sorbitan monostearate
About This Item
Produits recommandés
Agence
JPE
NF
Ph. Eur.
Niveau de qualité
Pression de vapeur
<10 hPa ( 20 °C)
Gamme de produits
EMPROVE® ESSENTIAL
Forme
viscous liquid
Technique(s)
API processing | nano-milling: suitable
pH
4,0-7.7 (25 °C, 50 g/L in H2O)
kinematic viscosity
170-320 cSt(40 °C)
Température de transition
flash point >149 °C
Densité
1.1 g/cm3 at 20 °C
Application(s)
liquid formulation
ophthalmics
pharma/biopharma processes
pharmaceutical
semi-solid formulation
solid formulation
solubility enhancement
Température de stockage
15-25°C
InChI
1S/C26H50O10/c1-2-3-4-5-6-7-8-9-10-11-24(30)34-19-18-31-20-22(32-15-12-27)26-25(35-17-14-29)23(21-36-26)33-16-13-28/h22-23,25-29H,2-21H2,1H3
Clé InChI
HMFKFHLTUCJZJO-UHFFFAOYSA-N
Description générale
As part of our Emprove® Program, our raw materials are offered with extensive documentation facilitating compliance of your pharma and biopharma product, full supply chain transparency and risk mitigation. Our SAFC® portfolio of high-quality products for biopharmaceutical and pharmaceutical formulation and production withstands strict quality control procedures and is produced according to applicable cGMP guidelines.
Application
Informations légales
Application
Produit(s) apparenté(s)
Code de la classe de stockage
10 - Combustible liquids
Classe de danger pour l'eau (WGK)
WGK 1
Point d'éclair (°F)
Not applicable
Point d'éclair (°C)
Not applicable
Certificats d'analyse (COA)
Recherchez un Certificats d'analyse (COA) en saisissant le numéro de lot du produit. Les numéros de lot figurent sur l'étiquette du produit après les mots "Lot" ou "Batch".
Déjà en possession de ce produit ?
Retrouvez la documentation relative aux produits que vous avez récemment achetés dans la Bibliothèque de documents.
Les clients ont également consulté
Articles
Medicine for children poses unique formulation challenges compared to adults. Consider developmental physiology and age specifics when designing pharmaceuticals. Quality issues can severely impact patient safety. Therefore, excipient quality, supplier selection, and supply chain security are crucial, particularly for pediatric formulations.
Medicine for children poses unique formulation challenges compared to adults. Consider developmental physiology and age specifics when designing pharmaceuticals. Quality issues can severely impact patient safety. Therefore, excipient quality, supplier selection, and supply chain security are crucial, particularly for pediatric formulations.
Medicine for children poses unique formulation challenges compared to adults. Consider developmental physiology and age specifics when designing pharmaceuticals. Quality issues can severely impact patient safety. Therefore, excipient quality, supplier selection, and supply chain security are crucial, particularly for pediatric formulations.
Medicine for children poses unique formulation challenges compared to adults. Consider developmental physiology and age specifics when designing pharmaceuticals. Quality issues can severely impact patient safety. Therefore, excipient quality, supplier selection, and supply chain security are crucial, particularly for pediatric formulations.
Contenu apparenté
Developing formulations specifically for infants and children is increasingly important. To help you master these challenges effectively, we offer an extensive portfolio of well-established high-quality excipients and APIs for pediatric pharmaceutical formulations that are proven in practice.
Developing formulations specifically for infants and children is increasingly important. To help you master these challenges effectively, we offer an extensive portfolio of well-established high-quality excipients and APIs for pediatric pharmaceutical formulations that are proven in practice.
Developing formulations specifically for infants and children is increasingly important. To help you master these challenges effectively, we offer an extensive portfolio of well-established high-quality excipients and APIs for pediatric pharmaceutical formulations that are proven in practice.
Developing formulations specifically for infants and children is increasingly important. To help you master these challenges effectively, we offer an extensive portfolio of well-established high-quality excipients and APIs for pediatric pharmaceutical formulations that are proven in practice.
Notre équipe de scientifiques dispose d'une expérience dans tous les secteurs de la recherche, notamment en sciences de la vie, science des matériaux, synthèse chimique, chromatographie, analyse et dans de nombreux autres domaines..
Contacter notre Service technique