72420
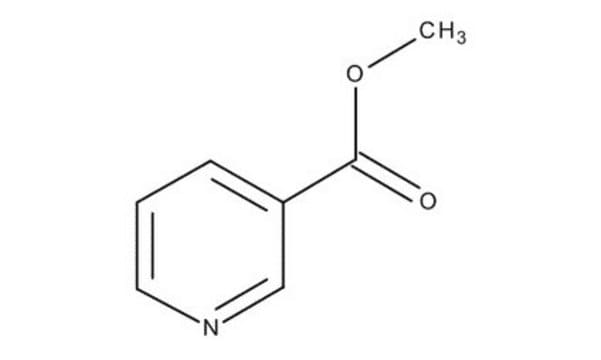
Methyl nicotinate
puriss., ≥99.0% (GC)
Synonym(s):
Nicotinic acid methyl ester
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Empirical Formula (Hill Notation):
C7H7NO2
CAS Number:
Molecular Weight:
137.14
Beilstein:
113951
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
biological source
synthetic
Quality Level
grade
puriss.
Assay
≥99.0% (GC)
form
powder or crystals (possibly with chunks)
color
white to faint yellow
bp
204 °C (lit.)
mp
39-42 °C
42-44 °C (lit.)
solubility
H2O: 0.1 g/mL, clear
SMILES string
COC(=O)c1cccnc1
InChI
1S/C7H7NO2/c1-10-7(9)6-3-2-4-8-5-6/h2-5H,1H3
InChI key
YNBADRVTZLEFNH-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
Methyl nicotinate (or nicotinic acid methyl ester) is used as a rubefacient for the relief of pains in muscles, tendons, and joints. It is also used in food as a flavoring agent.
Application
Methyl nicotinate can be used as a precursor:
- In the asymmetric synthesis of 1-azasugars (glycosidase inhibitors) for biomedical applications.
- In the total synthesis of ±-sesbanine.
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Selective fowler reductions: asymmetric total syntheses of isofagomine and other 1-azasugars from methyl nicotinate
Zhao G, et al.
Organic Letters, 3(2), 201-203 (2001)
Topical antirheumatic agents as hydroxyl radical scavengers
Billany MR, et al.
International Journal of Pharmaceutics, 124(2), 279-283 (1995)
A facile synthesis of ? -sesbanine via γ-addition of ketene silyl acetal with quaternized methyl nicotinate
Wada M, et al.
Tetrahedron Letters, 26(27), 3267-3270 (1985)
Stability of methylnicotinate in aqueous solution as utilized in the'niacin patch test'
Ross BM and Katzman M
BMC Research Notes, 1(1), 89-89 (2008)
Identification and quantification of methyl nicotinate in rice (Oryza sativa L.) by gas chromatography-mass spectrometry
RB Muralidhara, et al.
Food Chemistry, 105(2), 736-741 (2007)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service