08171
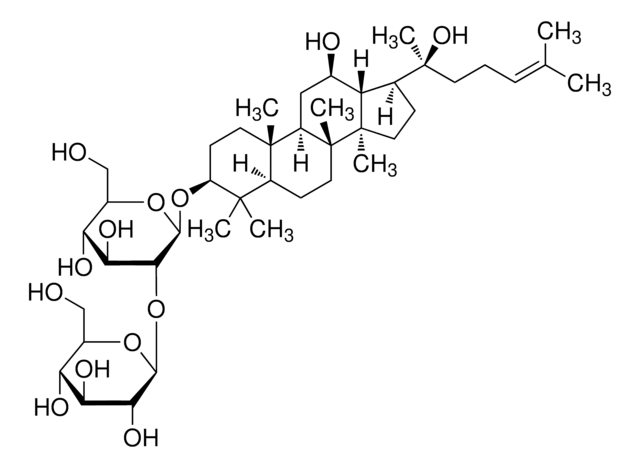
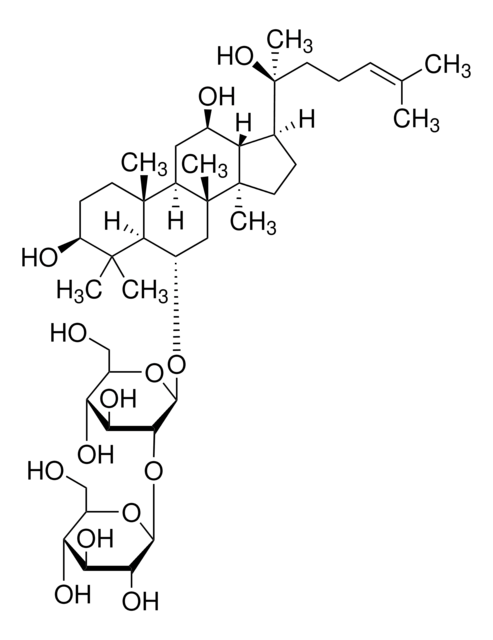
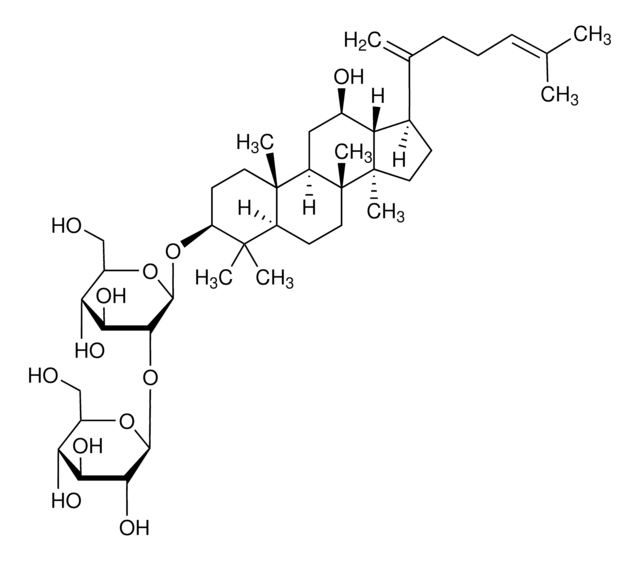
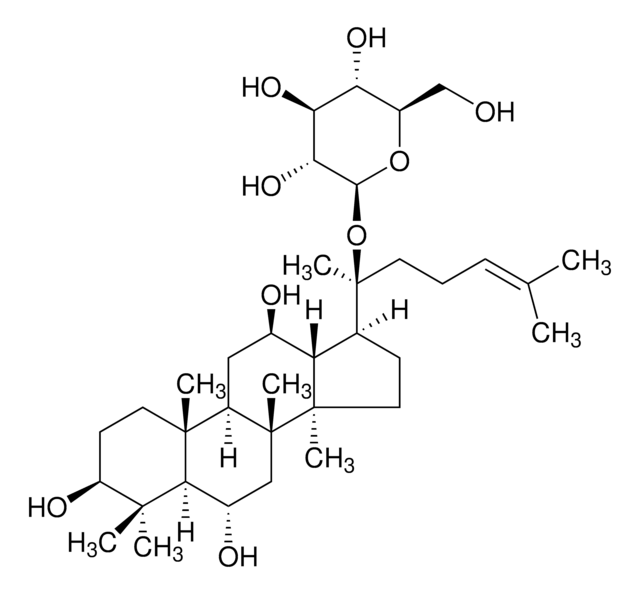
Ginsenoside Rg2
analytical standard
Synonyme(s) :
(3β,6α,12β)-3,12,20-Trihydroxydammar-24-en-6-yl 2-O-(6-deoxy-α-L-mannopyranosyl)-β-D-glucopyranoside, Chikusetsusaponin I, Panaxoside Rg2, Prosapogenin C2
About This Item
Produits recommandés
Qualité
analytical standard
Niveau de qualité
Pureté
≥94% (HPLC)
Durée de conservation
limited shelf life, expiry date on the label
Application(s)
food and beverages
Format
neat
Température de stockage
2-8°C
Chaîne SMILES
C[C@@H]1O[C@@H](O[C@@H]2[C@@H](O)[C@H](O)[C@@H](CO)O[C@H]2O[C@H]3C[C@]4(C)[C@H](C[C@@H](O)[C@@H]5[C@H](CC[C@@]45C)[C@@](C)(O)CC\C=C(/C)C)[C@@]6(C)CC[C@H](O)C(C)(C)[C@H]36)[C@H](O)[C@H](O)[C@H]1O
InChI
1S/C42H72O13/c1-20(2)11-10-14-42(9,51)22-12-16-40(7)28(22)23(44)17-26-39(6)15-13-27(45)38(4,5)35(39)24(18-41(26,40)8)53-37-34(32(49)30(47)25(19-43)54-37)55-36-33(50)31(48)29(46)21(3)52-36/h11,21-37,43-51H,10,12-19H2,1-9H3/t21-,22-,23+,24-,25+,26+,27-,28-,29-,30+,31+,32-,33+,34+,35-,36-,37+,39+,40+,41+,42-/m0/s1
Clé InChI
AGBCLJAHARWNLA-DQUQINEDSA-N
Vous recherchez des produits similaires ? Visite Guide de comparaison des produits
Catégories apparentées
Description générale
Conditionnement
Autres remarques
Mention d'avertissement
Warning
Mentions de danger
Conseils de prudence
Classification des risques
Acute Tox. 4 Oral
Code de la classe de stockage
11 - Combustible Solids
Classe de danger pour l'eau (WGK)
WGK 3
Point d'éclair (°F)
Not applicable
Point d'éclair (°C)
Not applicable
Choose from one of the most recent versions:
Déjà en possession de ce produit ?
Retrouvez la documentation relative aux produits que vous avez récemment achetés dans la Bibliothèque de documents.
Les clients ont également consulté
Articles
In this article we present several HPTLC applications and analytical standards for ginsenosides.
Notre équipe de scientifiques dispose d'une expérience dans tous les secteurs de la recherche, notamment en sciences de la vie, science des matériaux, synthèse chimique, chromatographie, analyse et dans de nombreux autres domaines..
Contacter notre Service technique