NIST8671
NISTmAb, Humanized IgG1k Monoclonal Antibody
NIST® SRM® 8671
About This Item
Recommended Products
antibody product type
primary antibodies
grade
certified reference material
form
liquid
manufacturer/tradename
NIST®
technique(s)
UV/Vis spectroscopy: suitable
liquid chromatography (LC): suitable
mass spectrometry (MS): suitable
format
matrix material
General description
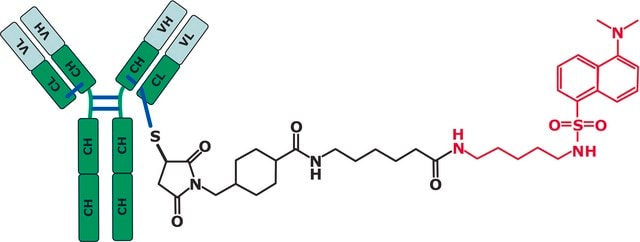
A unit of RM 8671 consists of one internal-threaded polypropylene cryovial containing 800 µL of 10 mg/mL NIST IgG1κ monoclonal antibody (NISTmAb) in 12.5 mmol/L L-histidine, 12.5 mmol/L L-histidine HCl (pH 6.0).
SRM 8671_Cert
SRM 8671_SDS
Application
- system suitability tests
- establishing method or instrument performance and variability
- comparing changing analytical test methods
- assisting in method qualification
Analysis Note
Other Notes
- RM 8671 is intended for research use.
- Details on expiration, storage, safety, usage, and source are provided in the NIST certificate.
- Information on biomaterials, disposal, and transport is available in the SDS.
Legal Information
Not finding the right product?
Try our Product Selector Tool.
related product
Storage Class Code
12 - Non Combustible Liquids
WGK
WGK 2
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Choose from one of the most recent versions:
Certificates of Analysis (COA)
Sorry, we don't have COAs for this product available online at this time.
If you need assistance, please contact Customer Support.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
An optimized peptide mapping protocol using NISTmAb as a model monoclonal antibody, shorter incubation times, and improved digestion buffer to demonstrate minimal artificial asparagine deamidation and methionine oxidation.
Compare columns in resolving medium-sized antibody fragments after digestion with DTT or IdeS using Reversed-Phase Chromatography for analysis.
Learn about the separation of several charge variants of two monoclonal antibodies, NISTmAb and Infliximab, using a polymeric cation exchange column and MS-compatible mobile phases.
This application note compares three different column chemistries from the BIOshell™ line of columns with superficially porous particles to evaluate their performance in peptide mapping.
Related Content
Discover our wide variety of products for intact mass analysis of monoclonal antibodies, including size-exclusion columns (SEC), ion exchange columns, reverse-phase columns, HPLC buffers, MALDI matrices and standards, high-purity solvents, reagents, tools for protein sample preparation, and certified reference materials.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service