448672
Poly(ethylene-co-acrylic acid)
acrylic acid 15 wt. %
Sign Into View Organizational & Contract Pricing
All Photos(2)
About This Item
Linear Formula:
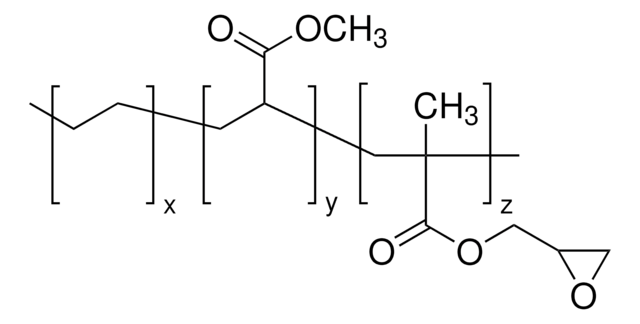
(CH2CH2)x[CH2CH(CO2H)]y
CAS Number:
MDL number:
UNSPSC Code:
12162002
PubChem Substance ID:
NACRES:
NA.23
Recommended Products
composition
acrylic acid, 15 wt. %
hardness
≤15 dmm (ASTM D 5, 25 °C)
viscosity
600 cP(140 °C, Brookfield)(lit.)
acid number
112‑130 mg KOH/g
transition temp
Tm (DSC) 87 °C (at peak)
density
0.93 g/mL at 25 °C
SMILES string
C=C.OC(=O)C=C
InChI
1S/C3H4O2.C2H4/c1-2-3(4)5;1-2/h2H,1H2,(H,4,5);1-2H2
InChI key
QHZOMAXECYYXGP-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Application
Processing and performance additive. Promotes crystallization of PET. Assists dispersion of additives in plastics.
Features and Benefits
Forms reversible ionic clusters (crosslinks). Promotes adhesion to various substrates, tougher, more chemically resistant and more transparent than parent acid copolymer.
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Ning Luo et al.
Colloids and surfaces. B, Biointerfaces, 50(2), 89-96 (2006-06-06)
Poly(ethylene-co-acrylic acid) (EAA) films were reacted with glycine, 12-aminododecanoic acid, aspartic acid, 5-aminoisophthalic acid, ethanolamine, diethylamine, dimethylamine, N-isopropylamine, and dimethylaminoethyleneamine to prepare EAA films with negatively charged, non-charged, hydrophilic, and hydrophobic functionalities. Attenuated total reflectance Fourier transform infrared spectroscopy, differential
Newton C Fawcett et al.
Langmuir : the ACS journal of surfaces and colloids, 20(16), 6651-6657 (2004-07-28)
Yoshimoto et al. [Anal. Chem. 2002, 74, 4306-4309] reported that a quartz crystal microbalance or QCM changed its response to sucrose solutions according to its angle of immersion. The effect was tentatively attributed to gravity-caused stress on the viscous interface
Siri Schauff et al.
Analytical chemistry, 79(21), 8323-8326 (2007-10-09)
The separation process in reversed-phase high-performance liquid chromatography employing C18 phases is mainly due to hydrophobic interactions. The separation of tocopherol isomers, exhibited by the C30 phases, however, is additionally driven by shape selectivity. This phenomenon is investigated by suspended-state
J Wegmann et al.
Analytical chemistry, 73(8), 1814-1820 (2001-05-08)
A new approach for the synthesis of long alkyl chain length stationary phases for use in reversed-phase liquid chromatography is described. Poly(ethylene-co-acrylic acid) copolymers (i.e., (-CH2CH2-)x[CH2CH(CO2H)-]y) with different levels of acrylic acid were covalently bonded to silica via glycidoxypropyl or
Marc David Grynbaum et al.
Journal of chromatography. A, 1156(1-2), 80-86 (2006-12-01)
The increased demand for chromatographic materials that are able to achieve a fast separation of large quantities of structure analogues is a great challenge. It is known that polymer based chromatographic materials have a higher loadability, compared to silica based
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service