911798
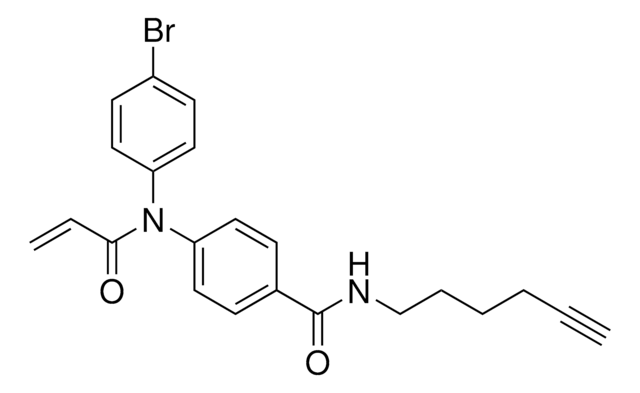
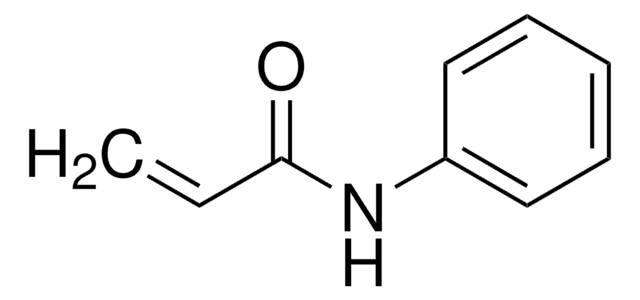
N-(4-Bromophenyl)-N-phenylacrylamide
≥95%
Synonym(s):
Electrophilic scout fragment, KB05, Scout fragment for targetable cysteine
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Empirical Formula (Hill Notation):
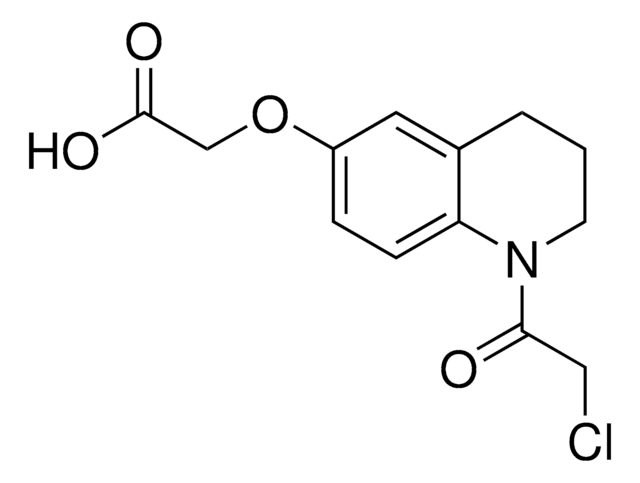
C15H12BrNO
CAS Number:
Molecular Weight:
302.17
UNSPSC Code:
12352200
NACRES:
NA.22
Recommended Products
Application
N-(4-Bromophenyl)-N-phenylacrylamide is a cysteine-reactive small-molecule fragment for chemoproteomic and ligandability studies for both traditionally druggable proteins as well as ″undruggable,″ or difficult-to-target, proteins. This fragment electrophile, or ″scout″ fragment, can be used alone in fragment-based covalent ligand discovery or incorporated into bifunctional tools such as electrophilic PROTAC® molecules for targeted protein degradation as demonstrated by the Cravatt Lab for E3 ligase discovery.
Other Notes
Legal Information
PROTAC is a registered trademark of Arvinas Operations, Inc., and is used under license
related product
Product No.
Description
Pricing
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Keriann M Backus et al.
Nature, 534(7608), 570-574 (2016-06-17)
Small molecules are powerful tools for investigating protein function and can serve as leads for new therapeutics. Most human proteins, however, lack small-molecule ligands, and entire protein classes are considered 'undruggable'. Fragment-based ligand discovery can identify small-molecule probes for proteins
Xiaoyu Zhang et al.
Nature chemical biology, 15(7), 737-746 (2019-06-19)
Ligand-dependent protein degradation has emerged as a compelling strategy to pharmacologically control the protein content of cells. So far, however, only a limited number of E3 ligases have been found to support this process. Here, we use a chemical proteomic
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service