S9381
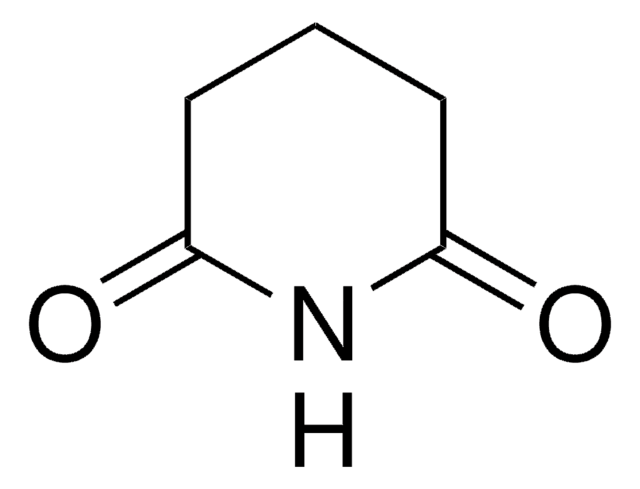
Succinimide
99.1%
Synonym(s):
2,5-Pyrrolidinedione
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Empirical Formula (Hill Notation):
C4H5NO2
CAS Number:
Molecular Weight:
99.09
Beilstein:
108440
EC Number:
MDL number:
UNSPSC Code:
12352209
PubChem Substance ID:
NACRES:
NA.26
Recommended Products
product name
Succinimide,
Assay
99.1%
form
powder or crystals
color
white to off-white
bp
285-290 °C (lit.)
mp
123-125 °C (lit.)
SMILES string
O=C1CCC(=O)N1
InChI
1S/C4H5NO2/c6-3-1-2-4(7)5-3/h1-2H2,(H,5,6,7)
InChI key
KZNICNPSHKQLFF-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Storage Class Code
11 - Combustible Solids
WGK
WGK 1
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
David Rennison et al.
Bioorganic & medicinal chemistry, 15(8), 2963-2974 (2007-02-27)
Norbormide [5-(alpha-hydroxy-alpha-2-pyridylbenzyl)-7-(alpha-2-pyridylbenzylidene)-5-norbornene-2,3-dicarboximide] (NRB, 1), an existing but infrequently used rodenticide, is known to be uniquely toxic to rats but relatively harmless to other rodents and mammals. A series of NRB-related analogues were prepared to investigate the structural features responsible for
Yutaka Sadakane et al.
Journal of chromatography. B, Analytical technologies in the biomedical and life sciences, 879(29), 3240-3246 (2011-04-08)
The major soluble eye lens protein, αA-crystallin, has a very long half-life. Thus, many post-translational modifications, including stereoinversion, have been found in its constituent amino acids. We determine the rates of β-linkage isomerization, which is the main reaction through the
Srinivasa Reddy Mothe et al.
The Journal of organic chemistry, 76(8), 2521-2531 (2011-03-15)
A one-pot, two-step method to prepare 3-halohydrofurans efficiently by TfOH-catalyzed hydroxylation/halocyclization of cyclopropyl methanols with H(2)O and N-halosuccinimide (NXS, X=1, Br, Cl) or Selectfluor is described. The reactions proceed rapidly under mild and operationally straightforward conditions with a catalyst loading
Yann Desfougères et al.
Biomacromolecules, 12(1), 156-166 (2010-12-21)
Protein chemical degradations occur naturally into living cells as soon as proteins have been synthesized. Among these modifications, deamidation of asparagine or glutamine residues has been extensively studied, whereas the intermediate state, a succinimide derivative, was poorly investigated because of
Ohgi Takahashi et al.
Chemistry & biodiversity, 7(6), 1630-1633 (2010-06-22)
The rapid racemization of aspartic acid (Asp) residues in peptides and proteins is due mainly to the succinimide intermediate. However, there should be another mechanism for Asp racemization without intermediacy of the succinimide. The direct H-atom abstraction from the C(alpha)-atom
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service