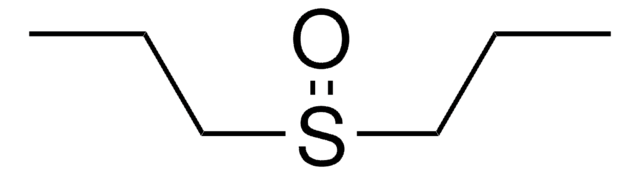P35405
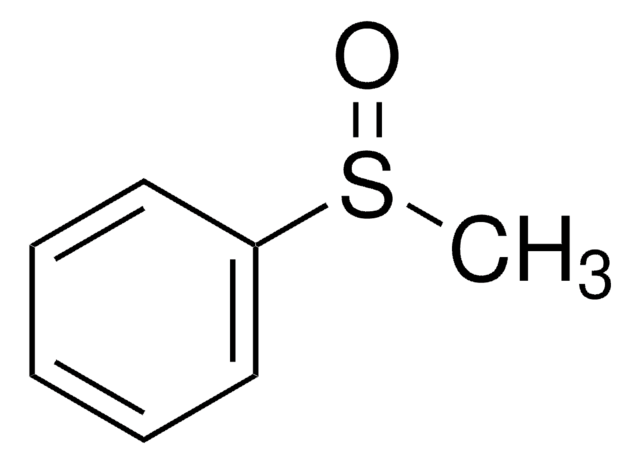
Diphenyl sulfoxide
96%
Synonym(s):
Phenyl sulfoxide
Sign Into View Organizational & Contract Pricing
All Photos(2)
About This Item
Linear Formula:
(C6H5)2SO
CAS Number:
Molecular Weight:
202.27
Beilstein:
1908444
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
96%
form
crystals
bp
206-208 °C/13 mmHg (lit.)
mp
69-71 °C (lit.)
SMILES string
O=S(c1ccccc1)c2ccccc2
InChI
1S/C12H10OS/c13-14(11-7-3-1-4-8-11)12-9-5-2-6-10-12/h1-10H
InChI key
JJHHIJFTHRNPIK-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Application
- Preparation of radiochemicals: Diphenyl sulfoxide plays a role in the synthesis of [(11)C]cyanide from [(11)C]methyl iodide, facilitating rapid and efficient production of radiochemicals for medical imaging applications (Kikuchi et al., 2022).
- Catalytic oxidation processes: The photocatalytic and catalytic oxidation of diphenyl sulphide to sulfoxide and sulfone was examined, highlighting the effectiveness of hydrogen peroxide and TiO2 polymorphs in optimizing chemical processes (Mikrut et al., 2022).
- Dielectric properties research: The study on dielectric properties of high organic sulfur coal highlighted the modeling of sulfur compounds, which could include diphenyl sulfoxide, enhancing our understanding of materials science in energy sectors (Cai et al., 2019).
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
S Yoshihara et al.
Drug metabolism and disposition: the biological fate of chemicals, 18(6), 876-881 (1990-11-01)
To evaluate the metabolic capacity of intact guinea pig liver under normoxic and hypoxic conditions, oxidative and reductive metabolism of diphenyl sulfoxide (DPSO) was studied by the nonrecirculating perfusion method in situ. DPSO was exclusively converted into diphenyl sulfone (DPSO2)
Martin A Fascione et al.
Chemistry (Weinheim an der Bergstrasse, Germany), 18(10), 2987-2997 (2012-02-02)
Sulfoxides are frequently used in organic synthesis as chiral auxiliaries and reagents to mediate a wide variety of chemical transformations. For example, diphenyl sulfoxide and triflic anhydride can be used to activate a wide range of glycosyl donors including hemiacetals
J A Kozlowski et al.
Bioorganic & medicinal chemistry letters, 10(20), 2255-2257 (2000-10-31)
Structure activity studies on [4-(phenylsulfonyl)phenyl]methylpiperazine led to the discovery of 4-cyclohexyl-alpha-[4-[[4-methoxyphenyl(S)-sufinyl]phenyl]-1-pi perazineacetonitrile, 1, an M2 selective muscarinic antagonist. Affinity at the cloned human M2 receptor was 2.7 nM; the M1/M2 selectivity is 40-fold.
S Yoshihara et al.
Archives of biochemistry and biophysics, 249(1), 8-14 (1986-08-15)
To characterize the properties of diphenyl sulfoxide (DPSO) as a new type of electron acceptor for guinea pig liver aldehyde oxidase (AO), we compared the kinetics of the reductions of DPSO and other classical electron acceptors such as O2 and
David Crich et al.
Organic letters, 8(5), 959-962 (2006-02-24)
The formation of sialic acid glycosides with a thiosialic acid derivative, diphenyl sulfoxide, and trifluoromethanesulfonic anhydride is reported. With an excess of diphenyl sulfoxide, glycal formation can be completely suppressed and excellent yields are obtained for coupling to a wide
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service