A97203
Azulene
99%
Synonym(s):
Bicyclo[5.3.0]decapentaene
Sign Into View Organizational & Contract Pricing
All Photos(2)
About This Item
Empirical Formula (Hill Notation):
C10H8
CAS Number:
Molecular Weight:
128.17
Beilstein:
969517
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
99%
form
crystals
bp
242 °C (lit.)
mp
98-100 °C (lit.)
SMILES string
c1ccc2cccc2cc1
InChI
1S/C10H8/c1-2-5-9-7-4-8-10(9)6-3-1/h1-8H
InChI key
CUFNKYGDVFVPHO-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
Packaging
Bottomless glass bottle. Contents are inside inserted fused cone.
Hazard Statements
Precautionary Statements
Hazard Classifications
Aquatic Chronic 2
Storage Class Code
11 - Combustible Solids
WGK
WGK 2
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Stefan Löber et al.
ChemMedChem, 4(3), 325-328 (2009-02-03)
Blue makes it happen: The non-uniform charge distribution of the blue colored azulene framework is highly suitable for the bioisosteric replacement of bicyclic heteroarene moieties. Showing an analogous binding mode as heterocyclic dopamine D4 receptor-selective lead compounds, the induction of
A F M Mustafizur Rahman et al.
Chemical communications (Cambridge, England), (10)(10), 1196-1198 (2008-03-01)
In spite of having small pi-conjugation systems, azulenes show large binding constants (10(4)-10(5)) to C(60) and C(70), which are larger than those of monoporphyrins and alternant aromatic hydrocarbons.
Ze-Yun Xiao et al.
Organic & biomolecular chemistry, 7(12), 2540-2547 (2009-06-09)
In this paper we report the synthesis and self-assembling behavior of new porphyrin-azulene-porphyrin and porphyrin-azulene conjugates. The porphyrin-azulene-porphyrin conjugate gelates a number of organic solvents, while the porphyrin-azulene conjugates form vesicles in a chloroform-methanol binary mixture. The structures of the
Atsuya Muranaka et al.
Journal of the American Chemical Society, 132(23), 7844-7845 (2010-05-27)
Azulenocyanine, having four azulene units fused to a tetraazaporphyrin skeleton, is a structural isomer of naphthalocyanine. We synthesized the first example of an azulenocyanine from 1,3-di-tert-butyl-5,6-dicyanoazulene. The macrocycle exhibits broad absorption over the visible and near-IR regions far beyond 1000
Timothy D Lash et al.
The Journal of organic chemistry, 77(5), 2368-2381 (2012-02-01)
A "2 + 2" strategy for synthesizing adj-dicarbaporphyrinoid systems has been developed. In a model study, an azulenylmethylpyrrole dialdehyde was condensed with a dipyrrylmethane in the presence of HCl, followed by oxidation with ferric chloride, to give a modest yield
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service



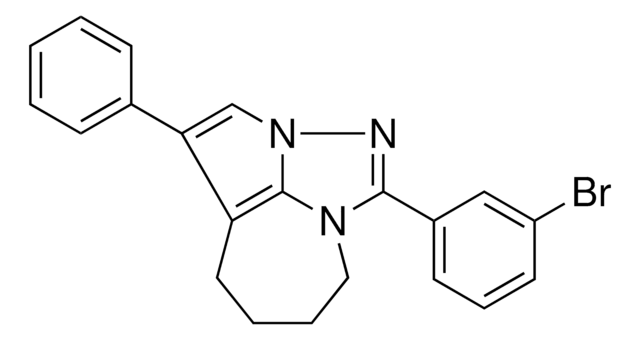
![(Ir[dF(CF3)ppy]2(dtbpy))PF6](/deepweb/assets/sigmaaldrich/product/structures/982/913/02dd8ddd-6deb-40a0-ab9b-07b18f1abb09/640/02dd8ddd-6deb-40a0-ab9b-07b18f1abb09.png)