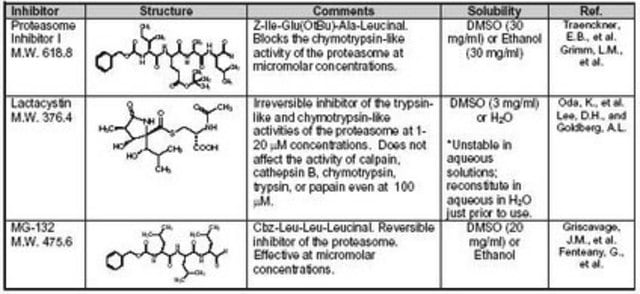
The molarity of a 25 mg/ml solution is 0.052 M, which is equivalent to 52 mM. This calculation is based on a substance with a molar mass of 475.62 g/mol, where a 1 M solution is prepared by dissolving 475.62 g in 1 liter.
M8699
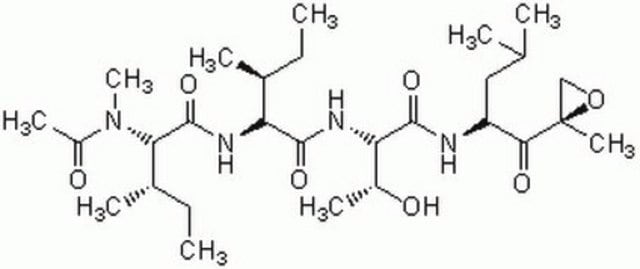
(R)-MG132
Synonym(s):
Z-L-Leu-D-Leu-L-Leu-al
About This Item
Recommended Products
Assay
≥98%
Quality Level
solubility
DMSO or DMF: 25 mg/mL
storage temp.
−20°C
SMILES string
CC(C)C[C@@H](C(N[C@H](C(N[C@H](C=O)CC(C)C)=O)CC(C)C)=O)NC(OCC1=CC=CC=C1)=O
InChI
1S/C26H41N3O5/c1-17(2)12-21(15-30)27-24(31)22(13-18(3)4)28-25(32)23(14-19(5)6)29-26(33)34-16-20-10-8-7-9-11-20/h7-11,15,17-19,21-23H,12-14,16H2,1-6H3,(H,27,31)(H,28,32)(H,29,33)/t21-,22-,23-/m0/s1
Application
Biochem/physiol Actions
Physical form
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
-
What is the molarity of a solution with a concentration of 25 mg/ml in DMSO?
1 answer-
Helpful?
-
-
Which cells are utilized to assess the bioactivity of Product No. M8699, also known as (R)-MG132?
1 answer-
The biological activity of product No. M8699, (R)-MG132 was measured using J558L multiple myeloma cells and EMT6 breast cancer cells.
Helpful?
-
Active Filters
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service