862126
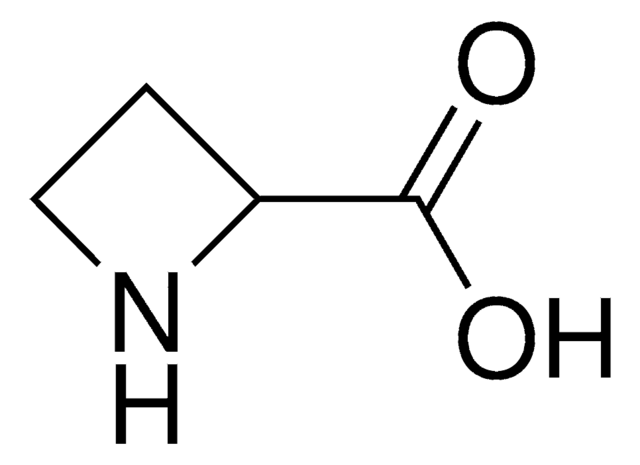
3,4-Dehydro-DL-proline
98%
Synonym(s):
(±)-2,5-Dihydro-1H-pyrrole-2-carboxylic acid, (±)-3-Pyrroline-2-carboxylic acid
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Empirical Formula (Hill Notation):
C5H7NO2
CAS Number:
Molecular Weight:
113.11
Beilstein:
471693
EC Number:
MDL number:
UNSPSC Code:
12352209
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
98%
form
crystals
reaction suitability
reaction type: solution phase peptide synthesis
mp
245 °C (dec.) (lit.)
application(s)
peptide synthesis
SMILES string
OC(=O)C1NCC=C1
InChI
1S/C5H7NO2/c7-5(8)4-2-1-3-6-4/h1-2,4,6H,3H2,(H,7,8)
InChI key
OMGHIGVFLOPEHJ-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
N S Fedarko et al.
Journal of bone and mineral research : the official journal of the American Society for Bone and Mineral Research, 10(11), 1705-1712 (1995-11-01)
Clinical studies indicate that as a group, osteogenesis imperfecta (OI) subjects are shorter than age- and sex-matched controls. Not only somatic growth, but also cellular growth appears to be impaired, and these may be related to defects in extracellular matrix
Maïté Vicré et al.
Plant physiology, 138(2), 998-1008 (2005-05-24)
Plant roots of many species produce thousands of cells that are released daily into the rhizosphere. These cells are commonly termed border cells because of their major role in constituting a biotic boundary layer between the root surface and the
G Xiao et al.
Molecular endocrinology (Baltimore, Md.), 11(8), 1103-1113 (1997-07-01)
Osteocalcin is a hormonally regulated calcium-binding protein made almost exclusively by osteoblasts. In normal cells, osteocalcin expression requires ascorbic acid (AA), an essential cofactor for osteoblast differentiation both in vivo and in vitro. To determine the mechanism of this regulation
XueLian Zhang et al.
Journal of experimental botany, 59(14), 4045-4058 (2008-10-22)
Extensins are cell wall basic glycoproteins with a polypeptide backbone that is extremely rich in hydroxyproline. In this paper, the function of extensins in embryo development was studied in Nicotiana tabacum. By using Western blot and immunohistochemistry, the extensin JIM20
D M Goli et al.
Carbohydrate research, 259(2), 219-241 (1994-06-17)
(2R,3R,4S)-2-Hydroxymethylpyrrolidine-3,4-diol (1,4-dideoxy-1,4-imino-D-ribitol) was synthesized in five steps from N-protected (2S)-3,4-dehydroproline methyl esters. The stereoselective reaction of osmium tetraoxide with dehydroproline derivatives gave high yields of (2S,3R,4S)-3,4-dihydroxyprolines (2,3-trans-3,4-cis-3,4-dihydroxy-L-prolines) accompanied by small amounts (< 15%) of the diastereomeric (2S,3S,4R)-3,4-dihydroxyprolines (2,3-cis-3,4-cis-3,4-dihydroxy-L-prolines). The mixture
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service