C1999
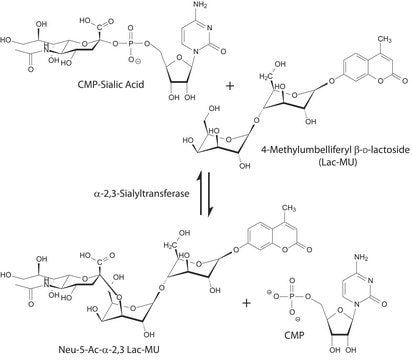
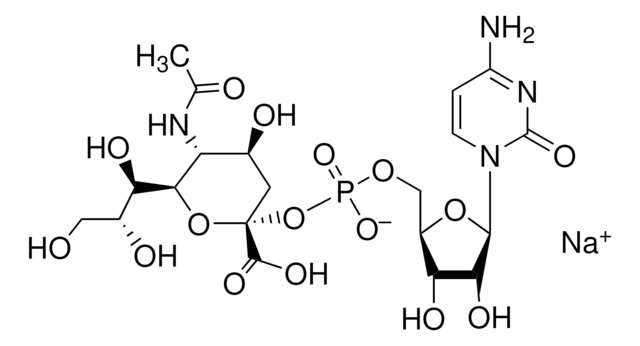
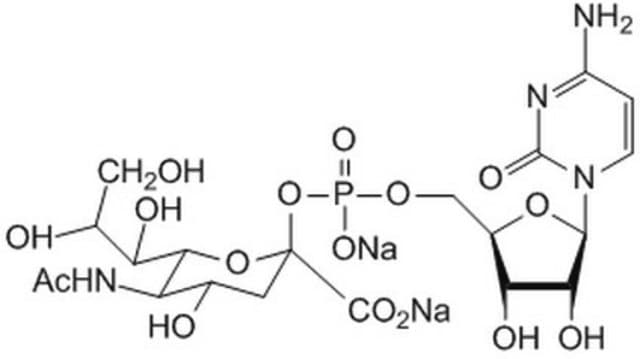
CMP-Sialic Acid Synthetase from Neisseria meningitidis group B
recombinant, expressed in E. coli BL21, ≥10 units/mg protein
Synonyme(s) :
CTP: N-Acylneuraminate cytidylyltransferase
About This Item
Produits recommandés
Produit recombinant
expressed in E. coli BL21
Niveau de qualité
Forme
lyophilized solid
Activité spécifique
≥10 units/mg protein
Poids mol.
26.0 kDa
Conditions d'expédition
dry ice
Température de stockage
−20°C
Vous recherchez des produits similaires ? Visite Guide de comparaison des produits
Description générale
Application
Actions biochimiques/physiologiques
Définition de l'unité
Forme physique
Remarque sur l'analyse
Code de la classe de stockage
11 - Combustible Solids
Classe de danger pour l'eau (WGK)
WGK 3
Point d'éclair (°F)
Not applicable
Point d'éclair (°C)
Not applicable
Certificats d'analyse (COA)
Recherchez un Certificats d'analyse (COA) en saisissant le numéro de lot du produit. Les numéros de lot figurent sur l'étiquette du produit après les mots "Lot" ou "Batch".
Déjà en possession de ce produit ?
Retrouvez la documentation relative aux produits que vous avez récemment achetés dans la Bibliothèque de documents.
Les clients ont également consulté
Articles
Explore tools for glycosyltransferase synthesis and modification of glycans, such as glycosyltransferases and nucleotide sugar donors.
Enzymatic glycosyltransferase specificity challenges the one enzyme-one linkage concept.
Understand sialic acid structure, function, signaling, and modifications. Easily find products for sialic acid research.
Notre équipe de scientifiques dispose d'une expérience dans tous les secteurs de la recherche, notamment en sciences de la vie, science des matériaux, synthèse chimique, chromatographie, analyse et dans de nombreux autres domaines..
Contacter notre Service technique