C2196
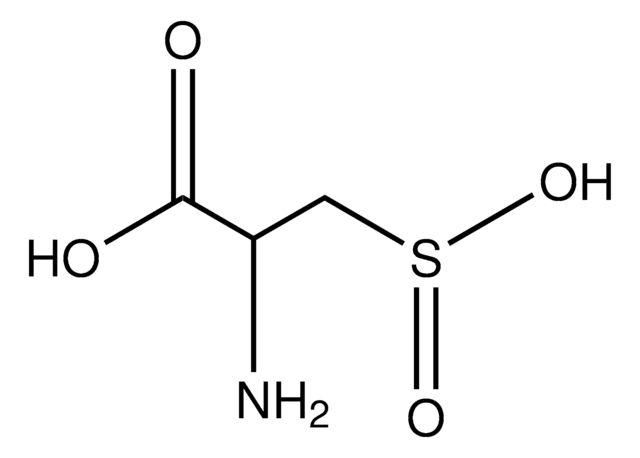
L-Cysteine S-sulfate
≥98% (TLC), suitable for ligand binding assays
Synonym(s):
S-Sulfo-L-cysteine
About This Item
Recommended Products
product name
L-Cysteine S-sulfate, ≥98% (TLC)
Quality Level
Assay
≥98% (TLC)
form
powder
technique(s)
ligand binding assay: suitable
color
white
storage temp.
−20°C
SMILES string
N[C@@H](CSS(O)(=O)=O)C(O)=O
InChI
1S/C3H7NO5S2/c4-2(3(5)6)1-10-11(7,8)9/h2H,1,4H2,(H,5,6)(H,7,8,9)/t2-/m0/s1
InChI key
NOKPBJYHPHHWAN-REOHCLBHSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
Biochem/physiol Actions
Cysteine is one of the functional amino acids that are associated with growth, reproduction, maintenance and immunity. Cysteine is a source of disulfide linkage in protein and is associated with sulfur transport. At physiological pH, cysteine undergoes rapid oxidation to form cystine. Reduced availability of cysteine or cystine, influences leukocyte metabolism. L-Cysteine serves as a precursor for the rate limiting step in glutathione synthesis that occurs in neurons. It donates inorganic sulfate for detoxification reactions. Therefore, L-cysteine might be associated with neuroprotection. It is found to obstruct the entry of heavy metal ions across the blood-brain barrier into the brain. Increased levels of L-cysteine might lead to neurotoxicity.
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service