51707
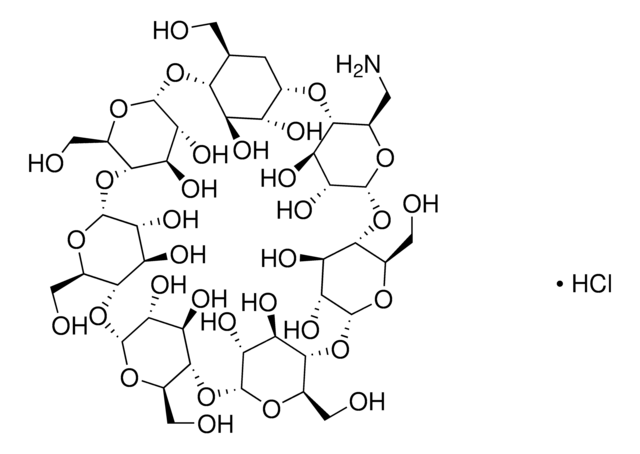
Heptakis(2,3,6-tri-O-methyl)-β-cyclodextrin
≥98.0%
Synonym(s):
TM-β-CD, TM‐β‐CyD, TRIMEB, 2,3,6-Tri-O-methyl-β-cyclodextrin, Trimethyl-β-cyclodextrin
About This Item
Recommended Products
Quality Level
Assay
≥98.0% (TLC)
≥98.0%
form
powder or crystals
optical activity
[α]25/D +157±4°, c = 1% in H2O
mp
170-178 °C (lit.)
solubility
H2O: 50 mg/mL, slightly turbid, colorless
storage temp.
2-8°C
SMILES string
COC[C@H]1O[C@@H]2O[C@@H]3[C@@H](COC)O[C@H](O[C@@H]4[C@@H](COC)O[C@H](O[C@@H]5[C@@H](COC)O[C@H](O[C@@H]6[C@@H](COC)O[C@H](O[C@@H]7[C@@H](COC)O[C@H](O[C@@H]8[C@@H](COC)O[C@H](O[C@H]1[C@H](OC)[C@H]2OC)[C@H](OC)[C@H]8OC)[C@H](OC)[C@H]7OC)[C@H](OC)[C@H]6OC)[C@H](OC)[C@H]5OC)[C@H](OC)[C@H]4OC)[C@H](OC)[C@H]3OC
InChI
1S/C63H112O35/c1-64-22-29-36-43(71-8)50(78-15)57(85-29)93-37-30(23-65-2)87-59(52(80-17)44(37)72-9)95-39-32(25-67-4)89-61(54(82-19)46(39)74-11)97-41-34(27-69-6)91-63(56(84-21)48(41)76-13)98-42-35(28-70-7)90-62(55(83-20)49(42)77-14)96-40-33(26-68-5)88-60(53(81-18)47(40)75-12)94-38-31(24-66-3)86-58(92-36)51(79-16)45(38)73-10/h29-63H,22-28H2,1-21H3/t29-,30-,31-,32-,33-,34-,35-,36-,37-,38-,39-,40-,41-,42-,43+,44+,45+,46+,47+,48+,49+,50-,51-,52-,53-,54-,55-,56-,57-,58-,59-,60-,61-,62-,63-/m1/s1
InChI key
DSDAICPXUXPBCC-MWDJDSKUSA-N
Looking for similar products? Visit Product Comparison Guide
General description
Application
- To investigate the crystal structure of its complexes with m-iodophenol, 4-biphenylacetic acid and (R)- and (S)-flurbiprofen by X-ray analysis.
- To study the candidature of its complex with vitamin A for potential application as a drug delivery system for ophthalmic applications by high sensitivity fluorescence spectrometry and high pressure liquid chromatography (HPLC).
- In the determination of analyte composition in commercial samples by HPLC coupled to mass spectrometry (MS).
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service