930083
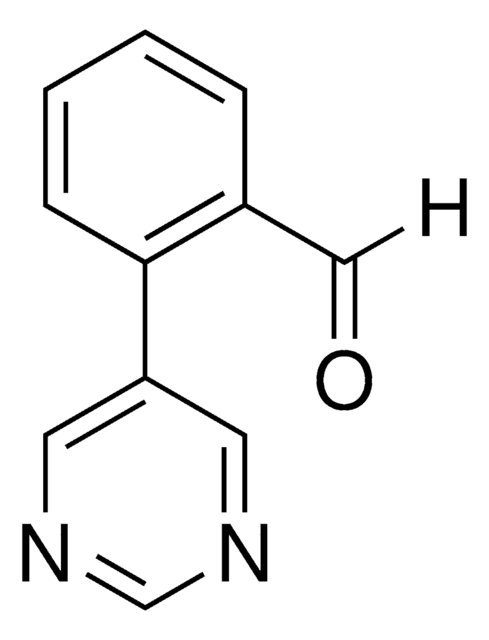
2-(Pyrimidin-5-yl)phenol
≥95%
Synonym(s):
Maiti-Bag-Dutta Auxiliary
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Empirical Formula (Hill Notation):
C10H8N2O
CAS Number:
Molecular Weight:
172.18
MDL number:
UNSPSC Code:
12352100
NACRES:
NA.21
Recommended Products
Application
2-(Pyrimidin-5-yl)phenol is a temporary directing group (TDG) to assist as a co-catalyst for metal catalyzed C-H functionalization. Often in C-H functionalization, an auxiliary compound is used to control site selectivity. These traditionally are covalently bonded to the compound of interest, and must subsequently be removed after functionalization, like a typical protecting group. To simplify the process of C-H functionalization, 2-Fluoro-6-(pyrimidin-5-yl)aniline is one of a series of temporary directing groups developed by Deb Maiti′s lab that promote site selectivity without the inclusion of additional synthetic steps.
2-(Pyrimidin-5-yl)phenol is an effective TDG for meta directed C-H functionalization of substitutes with a variety of electron withdrawing functional groups, with high selectivity.
2-(Pyrimidin-5-yl)phenol is an effective TDG for meta directed C-H functionalization of substitutes with a variety of electron withdrawing functional groups, with high selectivity.
related product
Product No.
Description
Pricing
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Sukdev Bag et al.
Journal of the American Chemical Society, 142(28), 12453-12466 (2020-06-05)
Controlling remote selectivity and delivering novel functionalities at distal positions in arenes are an important endeavor in contemporary organic synthesis. In this vein, template engineering and mechanistic understanding of new functionalization strategies are essential for enhancing the scope of such
Uttam Dutta et al.
Science (New York, N.Y.), 372(6543) (2021-05-15)
Transition metal-catalyzed aryl C-H activation is a powerful synthetic tool as it offers step and atom-economical routes to site-selective functionalization. Compared with proximal ortho-C-H activation, distal (meta- and/or para-) C-H activation remains more challenging due to the inaccessibility of these
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service