MABS1254
Anti-O-GlcNAc Antibody, clone CTD110.6
clone CTD110.6, from mouse
Synonym(s):
O-GlcNAc, beta-O-GlcNAc, O-Linked N-Acetylglucosamine, beta-O-linked N-acetylglucosamine
About This Item
Recommended Products
biological source
mouse
Quality Level
antibody form
purified immunoglobulin
antibody product type
primary antibodies
clone
CTD110.6, monoclonal
species reactivity (predicted by homology)
all
technique(s)
ELISA: suitable
immunoprecipitation (IP): suitable
western blot: suitable
isotype
IgMκ
target post-translational modification
glycosylation
Gene Information
human ... OGT(8473)
General description
Specificity
Immunogen
Application
Signaling
General Post-translation Modification
ELISA Analysis: A representative lot detected RNA polymerase II C-terminal domain (CTD) peptide (YSPTSPS) with a single O-GlcNAcylated serine or threonine, but not the corresponding unmodified peptide (Comer, F.I., et al. (2001). Anal. Biochem. 293(2):169-177).
Immunoprecipitation Analysis: A representative lot immunoprecipitated O-GlcNAcylated proteins from human pluripotent stem cells (hPSCs) (Maury, J.J., et al. (2013). Stem Cell Res. 11(2):926-937).
Immunoprecipitation Analysis: A representative lot immunoprecipitated O-GlcNAcylated proteins from HeLa cell extracts (Comer, F.I., et al. (2001). Anal. Biochem. 293(2):169-177).
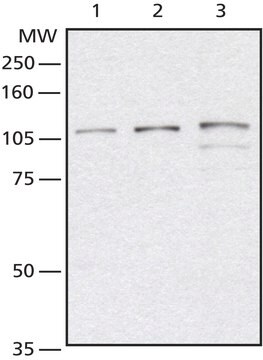
Western Blotting Analysis: A representative lot detected similar level of cellular O-GlcNAcylation in undifferentiated, differentiating and terminally differentiated human pluripotent stem cells (hPSCs) (Maury, J.J., et al. (2013). Stem Cell Res. 11(2):926-937).
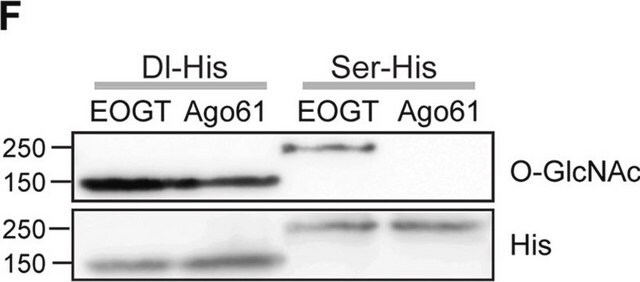
Western Blotting Analysis: A representative lot detected BSA-conjugated RNA polymerase II C-terminal domain (CTD) peptide (YSPTSPS) with beta-O-linked GlcNAc, but not alpha-O-linked GlcNAc, nor the corresponding unmodified peptide. The presence of GlcNAc, but not GalNAc, abolished the detection of the target bands (Comer, F.I., et al. (2001). Anal. Biochem. 293(2):169-177).
Western Blotting Analysis: A representative lot detected O-GlcNAcylated proteins in HeLa nuclear extract, as well as O-GlcNAcylated proteins purified from HeLa nuclear & cytosolic extract by wheat germ agglutinin (WGA) column. Antibody blocking by mmunogen peptide prior to immunoblotting abolished target bands detection (Comer, F.I., et al. (2001). Anal. Biochem. 293(2):169-177).
Western Blotting Analysis: A representative lot detected an upregulation of O-GlcNAcylated proteins in Jurkat cells treated with the glucosaminidase inhibitor PUGNAc and the hexosamine pathway intermediate glucosamine (Comer, F.I., et al. (2001). Anal. Biochem. 293(2):169-177).
Quality
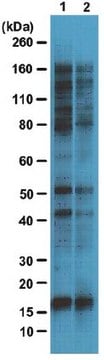
Western Blotting Analysis: 4.0 µg/mL of this antibody detected O-GlcNAcylated proteins in 10 µg of HeLa cell lysate.
Target description
Physical form
Storage and Stability
Other Notes
Disclaimer
Not finding the right product?
Try our Product Selector Tool.
Storage Class Code
10 - Combustible liquids
WGK
WGK 2
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service