All Photos(1)
About This Item
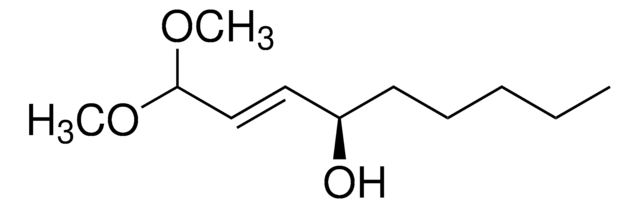
Empirical Formula (Hill Notation):
C4H6N4O
CAS Number:
Molecular Weight:
126.12
MDL number:
UNSPSC Code:
12352200
PubChem Substance ID:
Recommended Products
form
solid
SMILES string
NNC1=NC(=O)NC=C1
InChI
1S/C4H6N4O/c5-8-3-1-2-6-4(9)7-3/h1-2H,5H2,(H2,6,7,8,9)
InChI key
ZMYMWRORNQVELJ-UHFFFAOYSA-N
Other Notes
Please note that Sigma-Aldrich provides this product to early discovery researchers as part of a collection of unique chemicals. Sigma-Aldrich does not collect analytical data for this product. Buyer assumes responsibility to confirm product identity and/or purity. All sales are final.
NOTWITHSTANDING ANY CONTRARY PROVISION CONTAINED IN SIGMA-ALDRICH′S STANDARD TERMS AND CONDITIONS OF SALE OR AN AGREEMENT BETWEEN SIGMA-ALDRICH AND BUYER, SIGMA-ALDRICH SELLS THIS PRODUCT "AS-IS" AND MAKES NO REPRESENTATION OR WARRANTY WHATSOEVER WITH RESPECT TO THIS PRODUCT, INCLUDING ANY (A) WARRANTY OF MERCHANTABILITY; (B) WARRANTY OF FITNESS FOR A PARTICULAR PURPOSE; OR (C) WARRANTY AGAINST INFRINGEMENT OF INTELLECTUAL PROPERTY RIGHTS OF A THIRD PARTY; WHETHER ARISING BY LAW, COURSE OF DEALING, COURSE OF PERFORMANCE, USAGE OF TRADE OR OTHERWISE.
NOTWITHSTANDING ANY CONTRARY PROVISION CONTAINED IN SIGMA-ALDRICH′S STANDARD TERMS AND CONDITIONS OF SALE OR AN AGREEMENT BETWEEN SIGMA-ALDRICH AND BUYER, SIGMA-ALDRICH SELLS THIS PRODUCT "AS-IS" AND MAKES NO REPRESENTATION OR WARRANTY WHATSOEVER WITH RESPECT TO THIS PRODUCT, INCLUDING ANY (A) WARRANTY OF MERCHANTABILITY; (B) WARRANTY OF FITNESS FOR A PARTICULAR PURPOSE; OR (C) WARRANTY AGAINST INFRINGEMENT OF INTELLECTUAL PROPERTY RIGHTS OF A THIRD PARTY; WHETHER ARISING BY LAW, COURSE OF DEALING, COURSE OF PERFORMANCE, USAGE OF TRADE OR OTHERWISE.
Legal Information
Product of Synthonix
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
M Aida et al.
Biochemical and biophysical research communications, 153(2), 552-557 (1988-06-16)
The intrinsic properties of N4-aminocytosine, a base analogue of cytosine, are analyzed by an ab initio molecular orbital method. Relative stabilities of four possible isomeric structures of N4-aminocytosine are shown. The more stable isomer has the smaller dipole moment, so
N Nitta et al.
Nucleic acids symposium series, (6)(6), s43-s44 (1979-01-01)
N4-Aminocytosine reacted with acetone and acetaldehyde to form hydrazones that were readily revertible to the parent compound. With pyruvate, in contrast, it formed a stable hydrazone. By use of bromopyruvate, N4-aminocytosine was linked to glutathione.
K Matsumoto et al.
Mutation research, 268(1), 59-64 (1992-07-01)
N4-Aminocytidine is mutagenic in various organisms. In the cell, this cytidine analog is metabolized into N4-aminodeoxycytidine 5'-triphosphate, which will then be incorporated into DNA and mutation will result during the replication of the DNA. To prove that the N4-aminocytosine residue
Synthesis and properties of oligodeoxyribonucleotides containing a mutagenic base, N4-aminocytosine.
S Kuwazura et al.
Nucleic acids symposium series, (27)(27), 117-118 (1992-01-01)
Oligodeoxyribonucleotides containing a mutagenic base analog, N4-aminocytosine, 5'-AATTGC(am)AATT-3' and 5'-AATTAC(am)AATT-3' (C(am); N4-aminocytosine) were prepared by chemical modification of 5'-AATTGCAATT-3' and 5'-AATTACAATT-3', respectively. The values of Tm were 29 degrees C for 5'-AATTGC(am)AATT-3' and 32 degrees C for 5'-AATTGCAATT-3'. In contrast
A Nomura et al.
Mutation research, 177(2), 283-287 (1987-04-01)
N4-Aminocytidine induced mutation to 6-thioguanine resistance in Chinese hamster lung V79 cells in culture. Previous studies with experimental systems of in vitro DNA synthesis and of phage and bacterial mutagenesis have shown that this nucleoside analog induces base-pair transitions through
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service