H56800
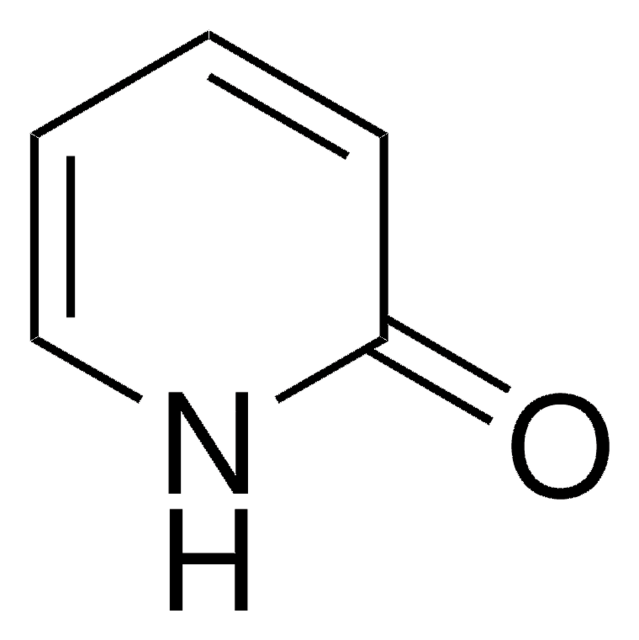
2-Hydroxypyridine
97%
Synonym(s):
2(1H)-Pyridone, 2-Pyridinol
Sign Into View Organizational & Contract Pricing
All Photos(2)
About This Item
Empirical Formula (Hill Notation):
C5H5NO
CAS Number:
Molecular Weight:
95.10
Beilstein:
105757
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
97%
bp
280-281 °C (lit.)
mp
105-107 °C (lit.)
SMILES string
Oc1ccccn1
InChI
1S/C5H5NO/c7-5-3-1-2-4-6-5/h1-4H,(H,6,7)
InChI key
UBQKCCHYAOITMY-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
Application
Catalyst for generating β-oxopropyl carbonates from cyclic carbonates and alcohols and in the aminolysis of a polyglutamate.
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 3 Oral
Storage Class Code
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
WGK
WGK 3
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Journal of the Chemical Society. Perkin Transactions 1, 1749-1749 (1993)
Ali A Abdul-Sater et al.
Journal of immunology (Baltimore, Md. : 1950), 195(1), 210-216 (2015-05-29)
IFNs, which transduce pivotal signals through Stat1 and Stat2, effectively suppress the replication of Legionella pneumophila in primary murine macrophages. Although the ability of IFN-γ to impede L. pneumophila growth is fully dependent on Stat1, IFN-αβ unexpectedly suppresses L. pneumophila
Polymer, 35, 2443-2443 (1994)
Mettu Ravinder et al.
Bioorganic & medicinal chemistry letters, 22(18), 6010-6015 (2012-08-18)
Twenty-six 2-pyridone derivatives (8a-8z), which are structurally analogous to amrinone and milrinone two important cardiotonic drugs, are synthesized and characterized. The synthesis of 2-pyridone derivatives involves addition, followed by cyclization between Baylis-Hillman acetates (7a-7k) and enamino esters or nitriles (3a-3e).
Borys Ośmiałowski et al.
Journal of molecular modeling, 17(10), 2491-2500 (2011-01-05)
The 2-[1H]-pyridone/2-hydroxypyridine tautomeric pair and its 6-substituted complexes have been studied with the use of DFT(M05) method. The intermolecular interaction energy has been calculated and discussed in the light of secondary interaction concept. The attractive secondary interactions of O/NH and
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service