344282
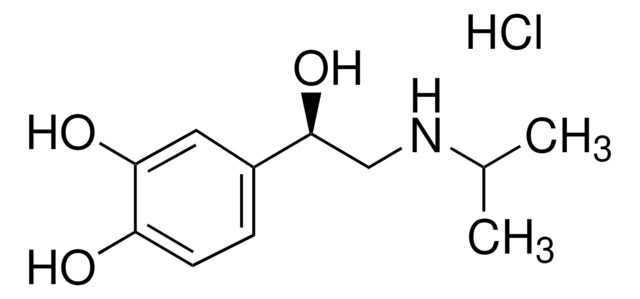
Forskolin
from Coleus forskohlii, ≥97% (HPLC), liquid, positive inotropic agent, Calbiochem
Synonym(s):
Forskolin, Coleus forskohlii in DMSO
About This Item
Recommended Products
product name
Forskolin, Coleus forskohlii in DMSO, The major cell-permeable diterpene isolated from the Indian plant Coleus forskohlii.
Quality Level
Assay
≥97% (HPLC)
form
liquid
manufacturer/tradename
Calbiochem®
storage condition
OK to freeze
desiccated (hygroscopic)
protect from light
shipped in
wet ice
storage temp.
−20°C
General description
Packaging
Warning
Physical form
Reconstitution
Other Notes
Noveen, A., et al. 1996. Biochem. Biophys. Res. Commun.219, 180.
Galli, C., et al. 1995. J. Neurosci.15, 1172.
Li, X., et al. 1995. Am. J. Physiol.269, C986.
Lomo, J., et al. 1995. J. Immunol.154, 1634.
Uneyama, H., et al. 1993. J. Biol. Chem.268, 168.
Laurenza, A., et al. 1989. Trends Pharmacol. Sci.10, 442.
Adashi, E.Y., and Resnick, C.E. 1986. J. Cell. Biochem.31, 217.
Seamon, K.B., and Daly, J.W. 1986. Adv. Cyclic Nucleotide Protein Phosphorylation Res.20, 1.
Huang, R., et al. 1982. Cyclic Nucleotide Res.8, 385.
Metzger, H., and Lindner, E. 1981. IRCS Med. Sci. Biochem. Cardiovasc. System Pharmacol.9, 99.
Legal Information
Storage Class Code
10 - Combustible liquids
WGK
WGK 2
Flash Point(F)
188.6 °F - (refers to pure substance)
Flash Point(C)
87 °C - (refers to pure substance)
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service